 Back
BackGeneral Chemistry Exam 1 Study Guide: Matter, Atoms, Molecules, and Chemical Reactions
Study Guide - Smart Notes

Chapter 1: Matter and Measurements
Physical States of Matter
The three physical states of matter—solid, liquid, and gas—are fundamental to understanding chemical substances. Each state has distinct properties:
Solid: Definite shape and volume; particles are closely packed and vibrate in place.
Liquid: Definite volume but no definite shape; particles are less tightly packed and can move past each other.
Gas: No definite shape or volume; particles are far apart and move freely.
Classification of Substances
Chemical substances can be classified as pure substances or mixtures:
Pure substances: Elements (single type of atom) or compounds (two or more elements chemically combined).
Mixtures: Homogeneous (uniform composition) or heterogeneous (non-uniform composition).
Chemical vs. Physical Changes
Understanding the difference between chemical and physical changes is essential:
Chemical change: Produces new substances (e.g., rusting of iron).
Physical change: Alters form but not composition (e.g., melting ice).
Intensive vs. Extensive Properties
Properties of matter are classified as:
Intensive properties: Independent of amount (e.g., density, boiling point).
Extensive properties: Dependent on amount (e.g., mass, volume).
SI Base Units and Metric Prefixes
The SI base units are fundamental for scientific measurement:
Length: meter (m)
Mass: kilogram (kg)
Temperature: kelvin (K)
Time: second (s)
Amount: mole (mol)
Metric prefixes modify base units by powers of ten. For example, 1 milligram (mg) = grams.
Temperature Conversions
Temperature can be converted between Celsius, Fahrenheit, and Kelvin using the following formulas:
Precision, Accuracy, and Significant Figures
Measurement quality is described by:
Precision: Consistency of repeated measurements.
Accuracy: Closeness to the true value.
Significant figures indicate the certainty in measurements. Rules for determining and reporting significant figures differ for multiplication/division and addition/subtraction.
Scientific Notation and Dimensional Analysis
Scientific notation expresses numbers as a product of a coefficient and a power of ten (e.g., ). Dimensional analysis uses conversion factors to change units and solve problems.
Chapter 2: Atoms, Molecules, and Ions
Subatomic Particles
Atoms are composed of three subatomic particles:
Proton: Positive charge, found in nucleus.
Neutron: Neutral, found in nucleus.
Electron: Negative charge, smallest mass, found outside nucleus.
Isotopes and Atomic Mass
Isotopes are atoms of the same element with different numbers of neutrons. Average atomic mass is calculated using:
Atomic Number and Mass
The atomic number gives the number of protons; atomic mass gives the sum of protons and neutrons. In a neutral atom, the number of electrons equals the number of protons.
Atomic Symbols and Ions
Atomic symbols show the element, atomic number, and mass. Ions are atoms with gained or lost electrons:
Cation: Positive charge, lost electrons.
Anion: Negative charge, gained electrons.
Empirical and Molecular Formulas
The empirical formula shows the simplest ratio of elements; the molecular formula shows the actual number of atoms in a molecule.
Periodic Table and Element Classification
The periodic table organizes elements by increasing atomic number and groups elements with similar properties. Elements are classified as metals or nonmetals.
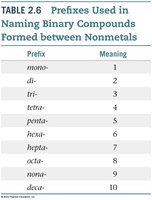
Naming Compounds: Prefixes for Binary Molecular Compounds
Binary molecular compounds (formed between nonmetals) use prefixes to indicate the number of atoms:
Prefix | Meaning |
|---|---|
mono- | 1 |
di- | 2 |
tri- | 3 |
tetra- | 4 |
penta- | 5 |
hexa- | 6 |
hepta- | 7 |
octa- | 8 |
nona- | 9 |
deca- | 10 |
Common Ions and Naming Rules
Monoatomic ions follow periodic patterns; polyatomic ions have specific names, formulas, and charges. Naming rules differ for:
Binary ionic compounds: Metal + nonmetal (e.g., NaCl).
Polyatomic ionic compounds: Contains polyatomic ions (e.g., Na2SO4).
Variable-charge ions: Transition metals; charge indicated by Roman numerals (e.g., Fe(III) chloride).
Molecular compounds: Use prefixes; only binary compounds (e.g., CO2 is carbon dioxide).
Chapter 3: Chemical Reactions and Reaction Stoichiometry
Law of Conservation of Mass
The law of conservation of mass states that mass is neither created nor destroyed in a chemical reaction.
Balancing Chemical Equations
Balancing equations ensures the same number of atoms of each element on both sides. Practice is essential for accuracy.
Types of Chemical Reactions
Combination: Two or more substances form one product.
Decomposition: One substance breaks into two or more products.
Combustion: Substance reacts with oxygen, producing energy and often CO2 and H2O.
Formula Weight and Percent Composition
Formula weight (FW) is the sum of atomic masses in a compound:
Percent composition is calculated as:
The Mole and Molar Mass
The mole (mol) represents particles (Avogadro's number). Molar mass is the mass of one mole of a substance, used as a conversion factor:
Stoichiometry and Limiting Reagents
Stoichiometry uses balanced equations to relate quantities of reactants and products. Molar ratios are derived from coefficients in the equation. The limiting reagent is the reactant that determines the maximum amount of product formed.
Theoretical Yield and Percent Yield
Theoretical yield is the maximum possible product; percent yield compares actual to theoretical:
Dimensional Analysis in Stoichiometry
Dimensional analysis is used to convert between mass, moles, and number of molecules, applying appropriate conversion factors.
Example: Stoichiometry Calculation
Given a balanced equation and starting mass of a reactant, calculate the mass of product formed:
Convert mass to moles using molar mass.
Use molar ratio from balanced equation.
Convert moles of product to mass.
Additional info: Practice and repetition are essential for mastering stoichiometry and chemical calculations.
