 Back
BackGeneral Chemistry Foundations for Organic Chemistry: Atomic Structure, Bonding, and Molecular Representation
Study Guide - Smart Notes

Ch.1 - Remembering General Chemistry (Part 1)
Concept: What is Organic Chemistry?
Organic chemistry is the study of molecules primarily composed of carbon and hydrogen, often found in biological systems. Organic molecules are defined by the presence of both carbon and hydrogen atoms. Molecules containing only carbon and hydrogen are called hydrocarbons.
Organic molecules: Contain both carbon and hydrogen.
Hydrocarbons: Organic molecules with only carbon and hydrogen.
Applications: Found in everyday products (e.g., hairspray ingredients) and biological processes (e.g., nerve gases).
Example: Identifying organic and hydrocarbon molecules from a product label.

Concept: Atomic Structure
The atom is the fundamental unit of matter. Its structure determines chemical properties and reactivity.
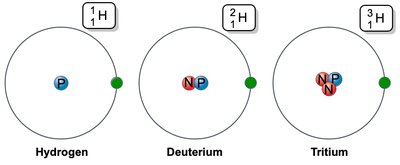
Atomic number: Number of protons in the nucleus.
Mass number: Sum of protons and neutrons.
Isotopes: Atoms with the same number of protons but different numbers of neutrons.
Electrons: Orbit the nucleus in defined regions called shells and orbitals.
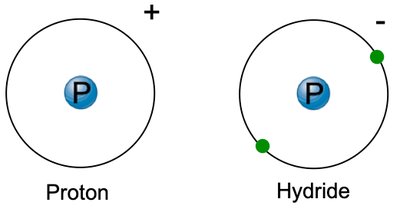
Ions: Atoms with unequal numbers of protons and electrons. Cations are positively charged; anions are negatively charged.
Concept: Electron Configuration Principles
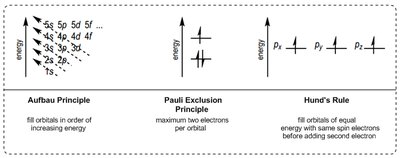
Electron configuration describes how electrons are distributed in an atom's orbitals. Three key principles govern this arrangement:
Aufbau Principle: Electrons fill orbitals in order of increasing energy.
Pauli Exclusion Principle: Each orbital can hold a maximum of two electrons with opposite spins.
Hund's Rule: Electrons fill orbitals of equal energy singly before pairing.
Concept: Quantum Mechanics and Wave Functions
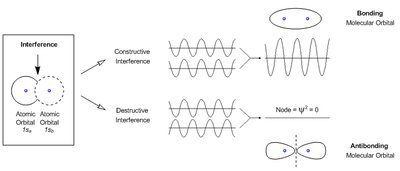
Quantum mechanics explains the dual nature of electrons as both particles and waves. The Heisenberg Uncertainty Principle states that an electron's position and momentum cannot both be precisely known. Wave functions describe the probability of finding an electron in a particular region, and their 3D plots are called atomic orbitals.
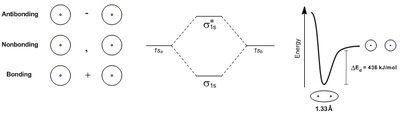
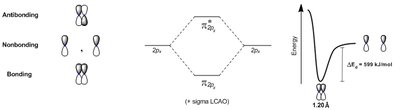
Wave function interference: Can be constructive (bonding) or destructive (antibonding).
Example: Formation of molecular orbitals in H2.
Concept: Molecular Orbital Theory
Molecular orbital (MO) theory describes how atomic orbitals combine to form molecular orbitals. The Linear Combination of Atomic Orbitals (LCAO) method uses addition and subtraction of orbitals to predict bonding and antibonding interactions.
Bonding orbitals: Formed by constructive overlap.
Antibonding orbitals: Formed by destructive overlap.
Energy diagrams: Show the relative energies of molecular orbitals.
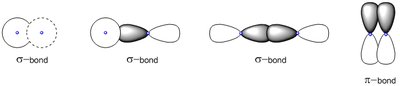
Concept: Sigma and Pi Bonds
Covalent bonds are classified as sigma (σ) or pi (π) bonds based on orbital overlap. Sigma bonds result from head-on overlap, while pi bonds result from side-to-side overlap.
Single bond: One sigma bond.
Double bond: One sigma and one pi bond.
Triple bond: One sigma and two pi bonds.
Bond length and strength: Triple bonds are shortest and strongest; single bonds are longest and weakest.
Single Bond | Double Bond | Triple Bond | |
|---|---|---|---|
Composition | 1 σ | 1 σ, 1 π | 1 σ, 2 π |
Free Rotation | Yes | No | No |
Length | Longest | Intermediate | Shortest |
Strength | Weakest | Intermediate | Strongest |
Concept: Octet Rule
Atoms are most stable when their valence shell contains eight electrons, achieving a noble gas configuration. This tendency is called the octet rule. Atoms may form bonds or possess lone pairs to satisfy the octet.
First-row elements: Prefer 2 octet electrons (H, He, Li).
Second-row elements: Prefer 8 octet electrons (C, N, O, F).
Exceptions: Be (4 electrons), B (6 electrons), third-row elements (expanded octets).
Concept: Bonding Preferences
Each element has a preferred number of bonds and lone pairs, determined by its group number. The most stable configuration is when the atom "owns" the correct number of electrons.
Valence electrons: Electrons in the outermost shell.
Bonding preference: Atoms prefer a specific number of bonds and lone pairs for stability.
Element | Group | Bonding Preference | Bonds | Lone Pairs |
|---|---|---|---|---|
Hydrogen (H) | 1A | 1 bond | 1 | 0 |
Beryllium (Be) | 2A | 2 bonds | 2 | 0 |
Boron (B) | 3A | 3 bonds | 3 | 0 |
Carbon (C) | 4A | 4 bonds | 4 | 0 |
Nitrogen (N) | 5A | 3 bonds | 3 | 1 |
Oxygen (O) | 6A | 2 bonds | 2 | 2 |
Fluorine (F) | 7A | 1 bond | 1 | 3 |
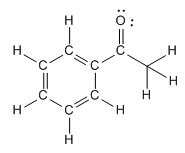
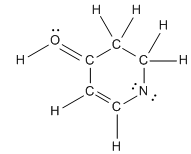
Concept: Formal Charges
Formal charge is the difference between the number of valence electrons an atom "owns" and its group number. The sum of all formal charges in a molecule is called the net charge.
Formula:
Application: Used to determine the most stable resonance structure.
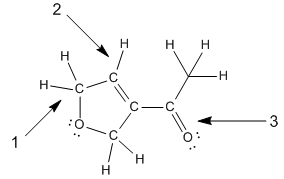
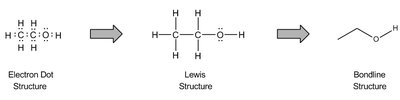
Concept: Skeletal Structure (Bondline Method)
The bondline (skeletal) method simplifies organic structure drawings. Carbons are implied at every vertex, hydrogens are implied to fill octets, and heteroatoms are shown explicitly. Formal charges are indicated when atoms do not satisfy their bonding preferences.
Implied atoms: Carbons and hydrogens are often not drawn.
Explicit atoms: Heteroatoms (O, N, etc.) and hydrogens on heteroatoms must be shown.
Conversion: Electron dot → Lewis structure → Bondline structure.
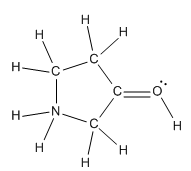
Practice and Application
Students are encouraged to practice determining atomic structure, electron configuration, formal charges, and converting between structural representations. These skills are foundational for understanding organic chemistry reactions and mechanisms.
Example: Assigning formal charges and net charges to line-angle structures.
Example: Identifying violations of the octet rule in molecules.
Additional info: These notes provide a bridge between general chemistry concepts and their application in organic chemistry, focusing on atomic structure, bonding, and molecular representation.
