 Back
BackGeneral Chemistry Foundations: Matter, Atoms, Molecules, and Solutions
Study Guide - Smart Notes

Chapter 1: Matter and Measurements
Matter and Its Composition
Matter is anything that occupies space and has mass. It is composed of atoms and molecules, which are the fundamental building blocks of chemistry.
Atoms: Smallest unit of matter, made up of protons, neutrons, and electrons.
Molecular element: Two or more of the same type of atom joined by a chemical bond (e.g., H2, O2).
Compounds: Two or more different atoms joined together by a chemical bond (e.g., CO2, H2O).
Scientific Method of Inquiry
Chemists use the scientific method to learn about the physical world. This involves making observations, forming hypotheses, developing laws, and constructing theories.
Law of Conservation of Mass: In a chemical reaction, matter is neither created nor destroyed.
Dalton's Atomic Theory: All matter is composed of small, indestructible particles called atoms.
Scientific Measurements and Notation
Measurements in chemistry require proper formatting and reporting to ensure clarity and reproducibility.
Scientific notation: Expresses numbers as a decimal and exponent (e.g., 5,283 = 5.283 x 103).
Reporting data: All digits except the last are certain; the last digit is estimated.
Significant Figures: Rules for addition/subtraction (smallest decimal place) and multiplication/division (smallest number of significant figures).
Rounding: If the digit after the last is 0-4, leave unchanged; if 5-9, round up.
Precision and Accuracy
Precision refers to how close a series of measurements are to one another, while accuracy measures how close a value is to the actual value.
Standard deviation:
Percent deviation:
Relative standard deviation (RSD):
Percent error:
SI Units and Metric Prefixes
Chemists use SI units based on the metric system for consistency.
Length: meter (m)
Mass: kilogram (kg)
Time: second (s)
Temperature: Kelvin (K)
Metric prefixes: Mega (106), Kilo (103), Milli (10-3), Micro (10-6), Nano (10-9), Pico (10-12)
Dimensional Analysis and Density
Dimensional analysis uses conversion factors to convert between units. Density is a key property used in conversions.
Density: (units: g/mL)
Energy Units and Temperature
Energy is measured in joules (J), calories (cal), and kilowatt-hours (kWh). Temperature conversions are essential in chemistry.
Kelvin:
Celsius: or
States of Matter and Types of Substances
Matter exists as solids, liquids, or gases. Substances can be pure (elements or compounds) or mixtures (homogeneous or heterogeneous).
Solids: Crystalline (ordered) or amorphous (disordered)
Liquids: Particles close but free to move
Gases: Particles far apart and move freely
Mixtures: Homogeneous (well mixed) or heterogeneous (distinct layers/parts)
Physical and Chemical Changes
Physical changes alter state or size without changing composition. Chemical changes result in new substances with different properties.
Physical separation techniques: Filtration, distillation, chromatography
Chapter 2: Elements, Atoms, and Ions
History and Atomic Theory
The concept of atoms originated with the Greeks and evolved through the work of many scientists. Dalton's Atomic Theory explained mass laws and the nature of atoms.
Mass Conservation: Atoms cannot be created or destroyed.
Definite Composition: Compounds have fixed ratios of elements by mass.
Multiple Proportions: Elements combine in ratios of small whole numbers.
Subatomic Particles
Atoms are composed of protons, neutrons, and electrons, each with distinct properties.
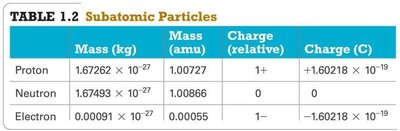
Proton: Positive charge, mass ≈ 1 amu
Neutron: Neutral, mass ≈ 1 amu
Electron: Negative charge, mass ≈ 0.00055 amu
Atomic Structure and Isotopes
Atoms are electrically neutral, with a nucleus containing protons and neutrons, surrounded by electrons. Isotopes are atoms of the same element with different numbers of neutrons.
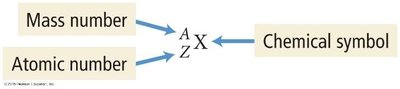
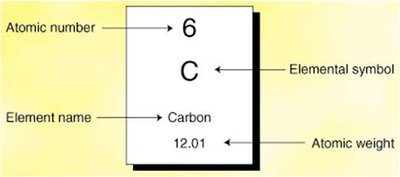
Atomic Number (Z): Number of protons
Mass Number (A): Number of protons + neutrons
Isotope notation: or X-A
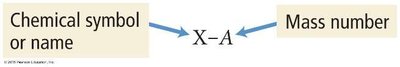
Periodic Table and Element Properties
The periodic table organizes elements by atomic number. Each element is defined by its number of protons, and can have varying numbers of neutrons (isotopes) and electrons (ions).
Cations: Positive ions (lost electrons)
Anions: Negative ions (gained electrons)
Groups: Columns with similar properties (e.g., alkali metals, halogens)
Compounds and Chemical Bonds
Compounds are formed by chemical bonds between atoms. Ionic bonds involve electron transfer; covalent bonds involve electron sharing.
Ionic compounds: Lattice of alternating ions
Covalent compounds: Individual molecules
Chemical formula: Indicates number and type of elements
Empirical formula: Simplest ratio of elements
Molecular formula: Actual number of atoms
Structural formula: Shows bonding arrangement
Naming Compounds
Compounds are named based on their composition and bonding type.
Type I: Metal + nonmetal, fixed charge
Type II: Metal + nonmetal, variable charge (use Roman numerals)
Type III: Two nonmetals (use prefixes for number of atoms)
Acids: Binary acids (hydro- + anion base + -ic acid), oxyacids (-ous for -ite, -ic for -ate)
Hydrocarbons: Alkanes (CnH2n+2), named by number of carbons
Formula Mass and Molar Mass
The formula mass (molecular mass, molar mass) is the sum of atomic masses in a compound, expressed in amu or g/mol.
Molar Mass:
Chapter 3: Solutions and Chemical Quantities
The Mole and Avogadro's Number
Counting individual atoms or molecules is impractical, so chemists use the mole as a counting unit. One mole contains 6.022 x 1023 particles (Avogadro's number).
Mole: Defined as exactly 12 grams of C-12 isotope
Molar Mass: Mass of one mole of a substance (g/mol)
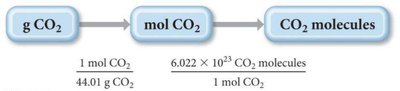
Converting Between Grams, Moles, and Molecules
To convert between mass, moles, and number of molecules, use the molar mass and Avogadro's number.
Step 1: Convert grams to moles using molar mass
Step 2: Convert moles to molecules using Avogadro's number
Empirical and Molecular Formulas
Empirical formulas show the simplest ratio of elements; molecular formulas show the actual number of atoms. The molecular formula is a multiple of the empirical formula.
Empirical formula calculation: Convert % composition to grams, then to moles, divide by smallest number of moles, adjust to whole numbers.
Molecular formula:
Solution Chemistry and Concentration
Solutions are homogeneous mixtures of two or more substances. The concentration of a solution describes the amount of solute present.
Solvent: Substance present in largest amount
Solute: Substance present in smaller amount
Molarity (M):
Molality (m):
Mole fraction:
Mass %:
ppm:
ppb:
Preparation and Dilution of Solutions
Solutions can be prepared by dissolving a known amount of solute in solvent. Dilutions are made using the equation .
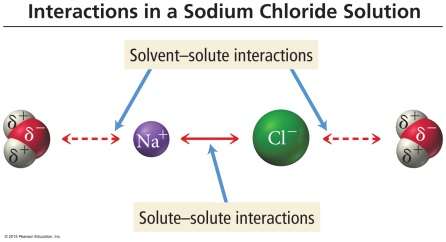
Interactions in Solutions
In aqueous solutions, interactions occur between solvent molecules, solute molecules, and between solvent and solute.
Concentration of Ions in Solution
The concentration of ions in solution can be calculated using the molarity of the compound and the mole ratio of ions.
Example: 6.00 M K2SO4 yields 12.00 M K+ and 6.00 M SO42-
Appendix: Subatomic Particle Properties
Particle | Mass (kg) | Mass (amu) | Charge (relative) | Charge (C) |
|---|---|---|---|---|
Proton | 1.67262 × 10-27 | 1.00727 | 1+ | +1.60218 × 10-19 |
Neutron | 1.67493 × 10-27 | 1.00866 | 0 | 0 |
Electron | 0.00091 × 10-27 | 0.00055 | 1– | –1.60218 × 10-19 |
