 Back
BackGeneral Chemistry I: Foundations of Matter, Measurement, and Atomic Theory
Study Guide - Smart Notes

Properties and States of Matter
Classification and States of Matter
Chemistry is the study of matter and energy. Matter is anything that has mass and occupies space. Matter can be classified by its state (solid, liquid, gas) and its properties.
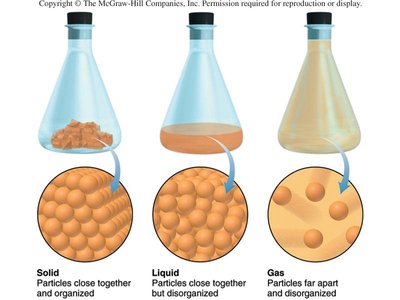
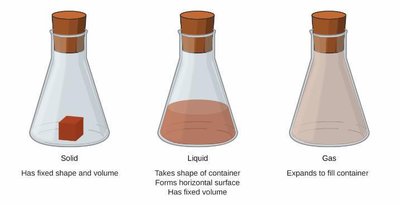
Solid: Particles are close together and organized; solids have a fixed shape and volume.
Liquid: Particles are close together but disorganized; liquids take the shape of their container and have a fixed volume.
Gas: Particles are far apart and disorganized; gases expand to fill their container.
Physical vs. Chemical Properties and Changes
Properties of matter can be classified as physical or chemical:
Physical Properties: Can be observed without changing the chemical composition (e.g., melting point, color, density).
Chemical Properties: Observed only when a substance undergoes a chemical change (e.g., flammability, reactivity).
Changes in matter are also classified as physical or chemical:
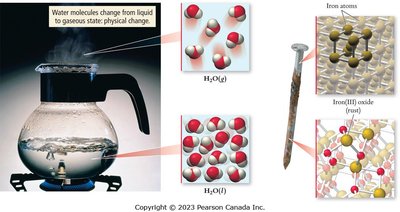
Physical Change: Alters the state or appearance but not the composition (e.g., melting, boiling).
Chemical Change: Alters the composition, resulting in new substances (e.g., rusting, combustion).
Classification of Matter: Pure Substances and Mixtures
Pure Substances vs. Mixtures
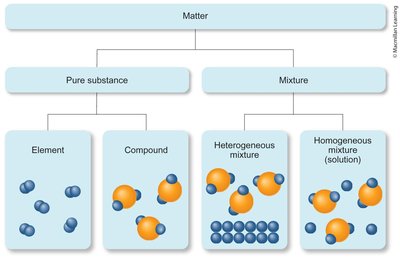
Matter can be classified as a pure substance or a mixture:
Pure Substance: Has a constant composition and distinct properties. Can be an element or a compound.
Mixture: Combination of two or more substances where each retains its own properties. Can be separated physically.
Types of Mixtures
Homogeneous Mixture (Solution): Uniform composition throughout (e.g., saltwater, tea with dissolved sugar).
Heterogeneous Mixture: Composition varies from one region to another (e.g., salad, iron filings in sand).



Measurement and Units in Chemistry
SI Units and Measurement
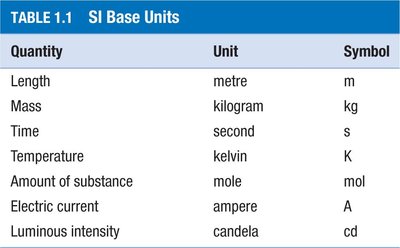
Measurements are fundamental to chemistry and must include a number, a unit, and an indication of uncertainty. The International System of Units (SI) is used globally for scientific measurements.
Quantity | Unit | Symbol |
|---|---|---|
Length | metre | m |
Mass | kilogram | kg |
Time | second | s |
Temperature | kelvin | K |
Amount of substance | mole | mol |
Electric current | ampere | A |
Luminous intensity | candela | cd |
Metric Prefixes
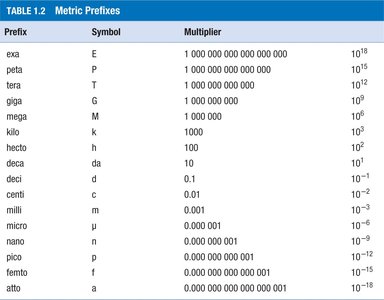
Prefixes are used to express very large or very small quantities conveniently.
Prefix | Symbol | Multiplier |
|---|---|---|
kilo | k | 103 |
centi | c | 10-2 |
milli | m | 10-3 |
micro | μ | 10-6 |
nano | n | 10-9 |
Scientific Notation
Scientific notation expresses very large or small numbers in the form , where and is an integer.
Example:
Example:
Measurement Uncertainty and Significant Figures
Uncertainty in Measurement
All measurements (except counting) have some degree of uncertainty. The uncertainty is reflected in the number of significant figures reported.
Significant Figures: All the digits in a measurement, including the uncertain last digit.
Rules for Counting Significant Figures:
All nonzero digits are significant.
Interior zeros are significant.
Leading zeros are not significant.
Trailing zeros are significant if after a decimal point or before a decimal point in a number with a decimal.

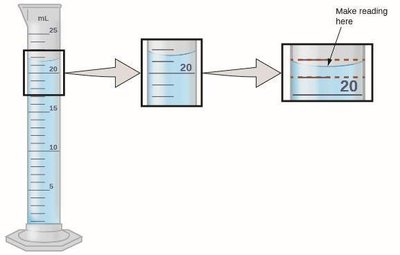



Precision and Accuracy
Precision: How close a series of measurements are to each other.
Accuracy: How close a measurement is to the true value.

Significant Figures in Calculations
Addition/Subtraction: Result has the same number of decimal places as the measurement with the fewest decimal places.
Multiplication/Division: Result has the same number of significant figures as the measurement with the fewest significant figures.

Dimensional Analysis and Unit Conversions
Dimensional Analysis (Factor-Label Method)
Dimensional analysis is used to convert between units using conversion factors. Always check that units cancel appropriately.
Write the quantity to be converted.
Multiply by conversion factors so units cancel, leaving the desired unit.
Check that the final units are correct.
Atomic Theory and Structure
Dalton’s Atomic Theory
Dalton’s Atomic Theory (early 19th century) laid the foundation for modern chemistry:
All matter is composed of indivisible atoms.
Atoms of a given element are identical in mass and properties.
Compounds are combinations of atoms of different elements in fixed ratios.
Chemical reactions involve rearrangement of atoms; atoms are not created or destroyed.
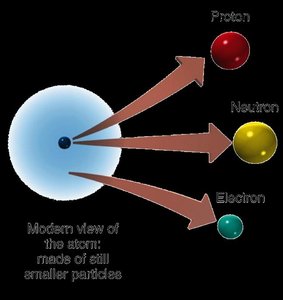
Subatomic Particles and Atomic Structure
Protons: Positively charged, located in the nucleus.
Neutrons: Neutral, located in the nucleus.
Electrons: Negatively charged, much lighter, found outside the nucleus.
Isotopes and Ions
Isotopes: Atoms of the same element with different numbers of neutrons (different mass numbers).
Ions: Atoms or molecules that have gained or lost electrons, resulting in a net charge.
The Periodic Table
Organization and Classification
The periodic table arranges elements by increasing atomic number and groups elements with similar properties into columns (groups or families).
Metals: Good conductors, malleable, ductile, tend to lose electrons.
Nonmetals: Poor conductors, can be solid, liquid, or gas, tend to gain electrons.
Metalloids: Exhibit properties of both metals and nonmetals.
Notable Groups
Group 1: Alkali metals (very reactive)
Group 2: Alkaline earth metals (fairly reactive)
Group 17: Halogens (very reactive nonmetals)
Group 18: Noble gases (very unreactive)
Sample Calculations and Practice
Mole Concept and Molar Mass
Mole (mol): The amount of substance containing entities (Avogadro’s number).
Molar Mass: The mass of one mole of a substance, numerically equal to the atomic or molecular mass in grams per mole.
Example Calculation:
Number of atoms in 2.26 g He: ; atoms
Additional info: For further practice, refer to the suggested textbook problems at the end of the lecture notes.
