 Back
BackGeneral Chemistry I: Introduction to Matter, Classification, and Properties
Study Guide - Smart Notes

Introduction to Chemistry
The Study of Chemistry
Chemistry is the scientific study of the properties, composition, and behavior of matter. It is a central science that connects and overlaps with biology, physics, environmental science, geology, and engineering. Understanding chemistry is essential for exploring the natural world and for advancements in technology, medicine, and environmental stewardship.
Chemistry: The study of the properties and behavior of matter.
Matter: Anything that has mass and occupies space.
Applications: Chemistry is foundational to biology, medicine, engineering, environmental sciences, agriculture, geology, and materials science.

Classification of Matter
States of Matter
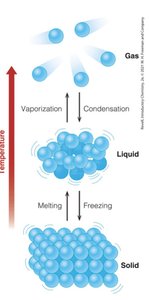
Matter exists in three primary physical states: solid, liquid, and gas. Each state is defined by the arrangement and movement of its particles.
Solids: Defined shape and volume; particles are closely packed with limited motion; incompressible.
Liquids: Defined volume but no fixed shape; particles have more freedom to move; incompressible.
Gases: No fixed shape or volume; particles move freely and are far apart; compressible.
Classification by Composition
Matter can be classified as pure substances or mixtures based on its composition and uniformity.
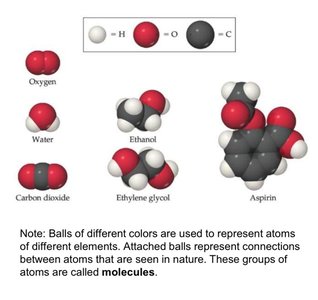
Pure Substances: Have a fixed composition and distinct properties. They are further divided into elements and compounds.
Mixtures: Consist of two or more substances physically combined; composition can vary.
Pure Substances
Elements: Made of only one type of atom; cannot be decomposed into simpler substances.
Compounds: Composed of two or more elements chemically combined in fixed ratios; can be decomposed into simpler substances.
Molecules: Groups of atoms bonded together, acting as a single unit.
Mixtures
Heterogeneous Mixtures: Not uniform in appearance; properties vary throughout the sample (e.g., salad, granite).
Homogeneous Mixtures (Solutions): Uniform in appearance and properties throughout (e.g., saltwater, air).
Properties of Matter
Physical and Chemical Properties
The properties of matter are used to describe and distinguish substances. They are classified as physical or chemical properties.
Physical Properties: Can be measured without changing the substance's identity (e.g., mass, volume, color, boiling point, density).
Chemical Properties: Can only be observed by changing the substance's identity (e.g., flammability, acidity, reactivity).
Intensive and Extensive Properties
Intensive Properties: Independent of the amount of substance (e.g., density, boiling point).
Extensive Properties: Depend on the amount of substance (e.g., mass, volume).
Changes in Matter
Physical and Chemical Changes
Changes in matter are classified as physical or chemical based on whether the composition of the substance changes.
Physical Change: Alters the form or appearance but not the composition (e.g., melting, boiling, dissolving).
Chemical Change: Alters the composition, resulting in new substances (e.g., burning, rusting).
Energy and Its Role in Chemistry
Types of Energy
Energy is the capacity to do work or transfer heat. It is involved in all physical and chemical changes.
Kinetic Energy: Energy of motion.
Potential Energy: Stored energy due to position or composition.
Heat Energy: Energy transferred due to temperature difference.
Physical and chemical changes involve energy changes, often in the form of heat:
Exothermic: Releases heat to the surroundings.
Endothermic: Absorbs heat from the surroundings.
Summary Table: Classification of Matter
Type | Description | Examples |
|---|---|---|
Element | One type of atom, cannot be decomposed | O2, Fe, Au |
Compound | Two or more elements, fixed ratio, can be decomposed | H2O, CO2 |
Homogeneous Mixture | Uniform composition throughout | Saltwater, air |
Heterogeneous Mixture | Non-uniform composition | Salad, granite |
Key Terms and Definitions
Atom: The smallest unit of an element that retains its chemical properties.
Molecule: A group of atoms bonded together.
Element: A pure substance made of only one kind of atom.
Compound: A pure substance composed of two or more elements in a fixed ratio.
Mixture: A physical blend of two or more substances.
Physical Property: A characteristic that can be observed without changing the substance's identity.
Chemical Property: A characteristic that can only be observed by changing the substance's identity.
Physical Change: A change that does not alter the chemical composition.
Chemical Change: A change that alters the chemical composition, forming new substances.
Energy: The capacity to do work or transfer heat.
Example: Classification of Water
Water (H2O) is a compound because it is composed of hydrogen and oxygen in a fixed ratio and can be decomposed into these elements.
Ice, liquid water, and steam are all physical states of the same compound.
Practice Problem
Classify each of the following as an element, compound, homogeneous mixture, or heterogeneous mixture:
Salt (NaCl)
Air
Iron filings
Salad
Answers: 1. Compound, 2. Homogeneous mixture, 3. Element, 4. Heterogeneous mixture
Further Study
Measurement and significant figures
Dimensional analysis
Units of measurement (SI units)
Additional info: For more advanced study, refer to the course textbook "Chemistry: The Central Science" (15th Edition) by Brown, LeMay, et al.
