 Back
BackGeneral Chemistry II: Kinetics, Solutions, and Thermodynamics Study Guide
Study Guide - Smart Notes

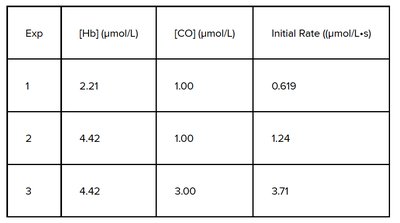
Q11. Determining the Rate Constant from Experimental Data
Background
Topic: Chemical Kinetics
This question tests your ability to determine the rate constant for a reaction using experimental data and the method of initial rates. You will need to deduce the rate law from the data and then solve for the rate constant.
Key Terms and Formulas
Rate Law:
k = rate constant (units depend on reaction order)
Initial Rate Method: Compare experiments where only one reactant concentration changes to determine reaction orders.
Step-by-Step Guidance
Write the general rate law for the reaction: .
Compare Experiments 1 and 2 (where [CO] is constant) to determine the order with respect to Hb. Set up the ratio of rates and concentrations to solve for m.
Compare Experiments 2 and 3 (where [Hb] is constant) to determine the order with respect to CO. Set up the ratio of rates and concentrations to solve for n.
Once you have the orders m and n, choose any experiment and plug the values for rate, [Hb], and [CO] into the rate law to solve for k.
Try solving on your own before revealing the answer!
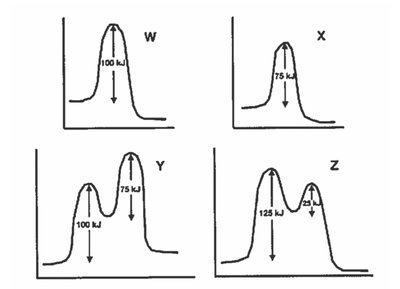
Q13. Analyzing Energy Profiles and Reaction Mechanisms
Background
Topic: Reaction Energy Diagrams and Mechanisms
This question asks you to interpret energy profile diagrams to determine information about activation energies, intermediates, and reaction spontaneity (exothermic/endothermic).
Key Terms and Concepts
Activation Energy (): The energy barrier that must be overcome for a reaction to proceed.
Intermediate: A species that appears in the mechanism but not in the overall reaction; corresponds to a valley between two peaks.
Transition State: The highest energy point along the reaction path; corresponds to a peak.
Exothermic Reaction: Products are lower in energy than reactants; overall energy change is negative.
Step-by-Step Guidance
Examine each diagram for the number of peaks (transition states) and valleys (intermediates).
Compare the energy of reactants and products to determine if the reaction is exothermic or endothermic.
Look at the activation energy for both forward and reverse reactions in each diagram.
Match the statements given in the question to the features you observe in the diagrams (e.g., number of intermediates, relative energies, etc.).
Try solving on your own before revealing the answer!
