 Back
BackGeneral Chemistry II: Thermochemistry and the First Law of Thermodynamics
Study Guide - Smart Notes

Thermochemistry and the First Law of Thermodynamics
Introduction to Thermodynamics and Thermochemistry
Thermodynamics is the branch of science that studies the relationships between heat and other forms of energy. Thermochemistry is a subfield of thermodynamics that focuses on the heat absorbed or evolved during chemical reactions. Understanding these concepts is fundamental for analyzing energy changes in chemical processes.
Thermodynamics: The science of the relationship between heat and other forms of energy.
Thermochemistry: The study of heat changes that accompany chemical reactions.
Energy and Its Units
Energy is the capacity to do work or produce heat. It exists in various forms, including kinetic, potential, and internal energy. The SI unit of energy is the joule (J), while the calorie (cal) is a commonly used non-SI unit in chemistry.
Kinetic Energy (EK): The energy of motion, given by the formula:
Potential Energy (EP): The energy due to position in a field of force (e.g., gravitational):
Internal Energy (U): The sum of the kinetic and potential energies of all particles in a system.
Total Energy:
Unit Conversions: (exact)

Law of Conservation of Energy
The law of conservation of energy states that energy can be converted from one form to another, but the total quantity of energy remains constant. This principle is foundational in all thermodynamic analyses.
Mathematical Expression:
Thermodynamic Systems and Surroundings
A thermodynamic system is the part of the universe under study, while the surroundings are everything else. The universe is thus composed of the system and its surroundings.
System: The specific part of the universe being studied (e.g., a reaction flask).
Surroundings: Everything outside the system.

Types of Systems
Open System: Both mass and energy can be exchanged with the surroundings (e.g., an open beaker).
Closed System: Only energy can be exchanged, not mass (e.g., a sealed flask).
Isolated System: Neither mass nor energy is exchanged (e.g., an insulated, closed container).
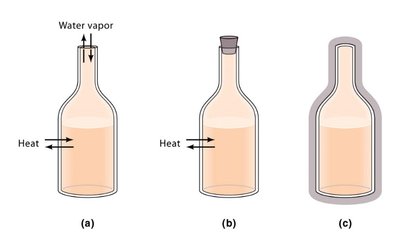
Work and Heat
Energy can be transferred between a system and its surroundings as work or heat.
Work (w): Energy transfer resulting from a force acting through a distance (e.g., moving a piston).
Heat (q): Energy transfer due to a temperature difference between the system and surroundings.
Sign Conventions:
Work is positive (+) when done on the system; negative (–) when done by the system.
Heat is positive (+) when absorbed by the system; negative (–) when evolved by the system.

State Functions and Path Functions
A state function is a property that depends only on the current state of the system, not on the path taken to reach that state. Internal energy (U) and enthalpy (H) are state functions, while work and heat are path functions.
State Function Example: Altitude is a state function; it depends only on the elevation, not the path taken.

The First Law of Thermodynamics
The first law of thermodynamics is a statement of energy conservation for thermodynamic systems. It relates the change in internal energy (ΔU) to heat (q) and work (w):
ΔU: Change in internal energy
q: Heat absorbed by the system
w: Work done on the system
Enthalpy and Enthalpy Changes
Enthalpy (H) is an extensive property that represents the heat content of a system at constant pressure. The change in enthalpy (ΔH) during a reaction is the heat absorbed or evolved at constant pressure.
Definition:
Change in Enthalpy:
Relationship to Internal Energy:
At constant pressure:
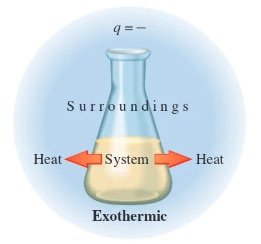
Endothermic vs. Exothermic Processes
Endothermic: Heat is absorbed (ΔH > 0); the system gains energy and the surroundings cool down.
Exothermic: Heat is released (ΔH < 0); the system loses energy and the surroundings warm up.
Pressure-Volume Work
When a reaction involves gases, the system may do work by expanding or contracting against external pressure. This is called pressure-volume (PV) work.
Formula:
Ideal Gas Law:
Work in terms of moles of gas:
Work is positive when the system is compressed (ΔV < 0), and negative when the system expands (ΔV > 0).
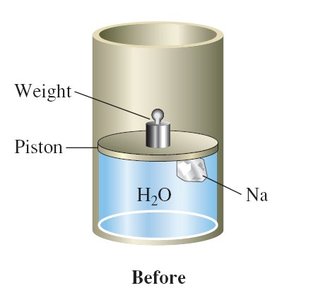
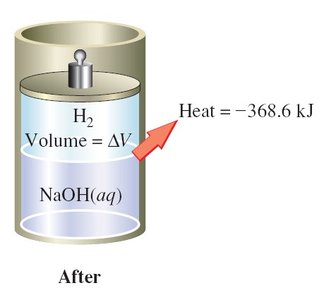
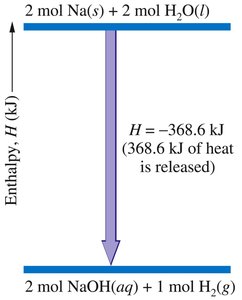
Thermochemical Equations
A thermochemical equation is a balanced chemical equation that includes the enthalpy change (ΔH) for the reaction as written. Phase labels are important, as the enthalpy change depends on the physical states of reactants and products.
Example:
Rules:
If the equation is multiplied by a factor, ΔH is multiplied by the same factor.
If the equation is reversed, the sign of ΔH is reversed.
Summary Table: Types of Thermodynamic Systems
System Type | Mass Exchange? | Energy Exchange? | Example |
|---|---|---|---|
Open | Yes | Yes | Beaker |
Closed | No | Yes | Sealed flask |
Isolated | No | No | Insulated, closed container |
Key Equations
End of Section Review
Understand the difference between heat and work as energy transfer mechanisms.
Be able to identify system, surroundings, and type of system in a given scenario.
Apply the first law of thermodynamics to calculate changes in internal energy.
Write and manipulate thermochemical equations, including correct use of ΔH and phase labels.
