Back
BackGeneral Chemistry Midterm Exam I: Study Notes and Worked Examples
Study Guide - Smart Notes

Gases and Gas Laws
Calculating Moles of a Gas Using the Ideal Gas Law
The Ideal Gas Law relates the pressure, volume, temperature, and amount of a gas. It is expressed as:
Equation:
P: Pressure (in kPa or atm)
V: Volume (in liters)
n: Number of moles
R: Universal gas constant ( J/mol·K)
T: Temperature (in Kelvin)
Example: To find the number of moles of NO(g) in a 275.0 mL container at 32.5°C and 75 kPa:
Convert volume to liters: L
Convert temperature to Kelvin: K
Plug values into the equation and solve for n:
*Additional info: This calculation demonstrates the use of the ideal gas law for determining the amount of gas present under non-standard conditions.*
Partial Pressures in Gas Mixtures (Dalton’s Law)
In a mixture of gases, each gas exerts a pressure independently of the others. The partial pressure of a gas is the pressure it would exert if it occupied the container alone.
Dalton’s Law of Partial Pressures:
For each gas:
Example: For 2.0 mol H2 and 1.0 mol N2 in a 22.4 L vessel at 298 K:
Calculate and separately using the ideal gas law.
Intermolecular Forces
Types of Intermolecular Interactions
Intermolecular forces are attractions between molecules that affect physical properties like boiling and melting points.
Hydrogen Bonding: Strong attraction between H and N, O, or F (e.g., H2O, NH2OH)
Dipole-Dipole: Attraction between polar molecules (e.g., NH2OH)
London Dispersion Forces: Present in all molecules, especially nonpolar ones (e.g., CBr4)
Examples:
NH2OH: Hydrogen bonding, dipole-dipole, dispersion
CBr4: Dispersion only
H2O: Hydrogen bonding, dipole-dipole, dispersion
Thermochemistry
Calculating Heat for Temperature Change
The heat required to change the temperature of a substance is given by:
Equation:
q: Heat (in Joules)
m: Mass (in kg)
c: Specific heat capacity (J/kg·K)
\Delta T: Change in temperature (K or °C)
Example: To heat 300.0 g (0.300 kg) of water from 20.0°C to 100.0°C:
°C
Enthalpy of Reaction and Formation
The enthalpy of formation () is the heat change when one mole of a compound forms from its elements. The reaction enthalpy change () can be calculated using enthalpies of formation:
Equation:
Example: Calculating the reaction enthalpy for burning TNT using its value.
Spontaneity and Thermodynamics
Criteria for Spontaneity
A chemical reaction is spontaneous at constant pressure and temperature if the Gibbs free energy change () is negative:
Equation:
If , the reaction is spontaneous.
Temperature for Spontaneity
To estimate the temperature at which a reaction becomes spontaneous:
Set and solve for :
Phase Changes and Vapor Pressure
Clapeyron-Clausius Equation
The Clapeyron-Clausius equation relates vapor pressure and temperature for phase changes:
Used to estimate boiling points and vapor pressures at different temperatures.
Solutions and Solubility
Solvent Choice and Solubility Principles
Solubility depends on the principle "like dissolves like":
Ionic and polar substances dissolve best in polar solvents (e.g., water).
Nonpolar substances dissolve best in nonpolar solvents (e.g., benzene).
Examples:
KCl: Water (polar)
CCl4: Benzene (nonpolar)
CH3COOH: Water (polar, but also some solubility in benzene due to nonpolar tail)
Molality Calculation
Molality (m) is the number of moles of solute per kilogram of solvent:
Example: Dissolving 25.0 g NaCl in 500.0 g water:
Find moles of NaCl:
Convert solvent to kg: kg
Calculate molality.
Bonus: Balancing Equations and Thermodynamic Feasibility
Balancing Chemical Equations
Balancing equations ensures the same number of each atom on both sides. For reactions involving rocket fuels, balance the equation before calculating thermodynamic properties.
Assessing Rocket Fuel Feasibility
To determine if a fuel is suitable, check if its oxidation is spontaneous at high temperature (e.g., 2000 K) using values. If , the reaction is thermodynamically favorable.
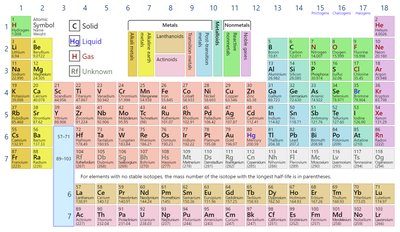
Reference: Periodic Table of the Elements
The periodic table organizes elements by atomic number and properties. It is essential for identifying element symbols, atomic masses, and chemical families.