 Back
BackGeneral Chemistry Review: Orbitals, Bonding, Electron Configurations, and Resonance
Study Guide - Smart Notes

Q1. What is the best description of an orbital?
Background
Topic: Atomic Structure & Quantum Chemistry
This question tests your understanding of what an atomic orbital is and how it relates to the probability of finding electrons in an atom.
Key Terms:
Orbital: A region in an atom where there is a high probability of finding an electron.
Electron cloud: The area around the nucleus where electrons are likely to be found.
Step-by-Step Guidance
Recall that electrons do not follow fixed paths (like planets), but are found in regions of space called orbitals.
Consider the quantum mechanical model, which describes orbitals as probability distributions rather than physical paths.
Eliminate options that mention protons or neutrons, as orbitals are specifically related to electrons.
Focus on the definition that emphasizes probability and the spatial region around the nucleus.
Try solving on your own before revealing the answer!
Q2. Which of the following shows a picture of a p-orbital?
Background
Topic: Atomic Orbitals & Their Shapes
This question tests your ability to recognize the visual representation of a p-orbital compared to s- and d-orbitals.
Key Terms:
s-orbital: Spherical shape
p-orbital: Dumbbell shape
d-orbital: Cloverleaf shape
Step-by-Step Guidance
Review the typical shapes of s, p, and d orbitals.
Identify the dumbbell-shaped orbital among the provided images.
Compare the shapes: spherical (s), dumbbell (p), and cloverleaf (d).
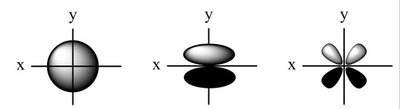
Try solving on your own before revealing the answer!
Q3. For a given shell, how many p orbitals exist?
Background
Topic: Quantum Numbers & Orbital Types
This question tests your understanding of the number of p orbitals in a shell, based on quantum numbers.
Key Terms:
p-orbitals: Orbitals with angular momentum quantum number
Each p subshell contains three orbitals: , ,
Step-by-Step Guidance
Recall that the p subshell corresponds to .
For , the magnetic quantum number can be , giving three orbitals.
List the three p orbitals: , , .
Try solving on your own before revealing the answer!
Q4. Use the following diagram to determine which orbital experiences the highest effective nuclear charge (Zeff)? Which orbital is lowest in energy? Briefly explain how you used the diagram to determine your answer.
Background
Topic: Radial Distribution & Effective Nuclear Charge
This question tests your ability to interpret radial probability graphs and relate them to effective nuclear charge and orbital energy.
Key Terms and Formulas:
Effective nuclear charge (): The net positive charge experienced by an electron in a multi-electron atom.
Radial probability: Likelihood of finding an electron at a certain distance from the nucleus.
Penetration: How close an orbital gets to the nucleus; greater penetration means higher .
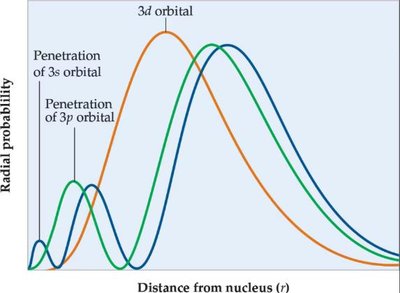
Step-by-Step Guidance
Examine the diagram and identify which orbital's probability distribution is closest to the nucleus.
Recall that orbitals with greater penetration (closer to the nucleus) experience higher .
Compare the energy levels: orbitals closer to the nucleus are generally lower in energy.
Use the labels on the graph to match each curve to its corresponding orbital (3s, 3p, 3d).
Try solving on your own before revealing the answer!
Q5. Write the ground state electron configuration for each of the following using the noble gas convention. How many unpaired electrons are in each?
Background
Topic: Electron Configurations & Unpaired Electrons
This question tests your ability to write electron configurations using noble gas notation and determine the number of unpaired electrons.
Key Terms and Formulas:
Noble gas notation: Use the symbol of the nearest noble gas to abbreviate the electron configuration.
Unpaired electrons: Electrons in orbitals that are not paired with another electron of opposite spin.
Step-by-Step Guidance
Identify the atomic number for each element (C, F, Ga, Bi, Pb).
Write the electron configuration using the noble gas abbreviation.
Fill the orbitals according to the Aufbau principle, Hund's rule, and the Pauli exclusion principle.
Count the number of unpaired electrons in the outermost orbitals.
Try solving on your own before revealing the answer!
