 Back
BackGeneral Chemistry Study Guidance: Bonding, Structure, and Atomic Properties
Study Guide - Smart Notes

Q16. Draw the 3 resonance structures of SO3.
Background
Topic: Resonance Structures and Lewis Structures
This question tests your understanding of resonance in molecules, specifically how electrons can be delocalized in a molecule like sulfur trioxide (SO3), resulting in multiple valid Lewis structures.
Key Terms:
Resonance: The concept that some molecules can be represented by two or more valid Lewis structures, called resonance structures, which differ only in the placement of electrons.
Lewis Structure: A diagram showing the arrangement of atoms and electrons in a molecule.
Step-by-Step Guidance
Draw the basic skeleton of SO3 with sulfur in the center and three oxygens around it.
Assign one double bond and two single bonds between sulfur and oxygen, then distribute lone pairs to satisfy the octet rule for each oxygen.
Move the double bond to each oxygen in turn, creating three distinct resonance structures.
Ensure that each structure has the same overall number of electrons and that formal charges are minimized.
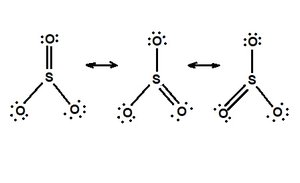
Try solving on your own before revealing the answer!
Q17. Complete the molecular orbital diagrams for diatomic fluorine and calculate the bond order for F2.
Background
Topic: Molecular Orbital Theory
This question tests your ability to interpret molecular orbital diagrams and use them to determine the bond order in diatomic molecules like F2.
Key Terms and Formulas:
Molecular Orbital (MO): Orbitals formed from the combination of atomic orbitals when atoms bond.
Bond Order Formula:
Step-by-Step Guidance
Draw or analyze the molecular orbital diagram for F2, showing the arrangement of electrons in bonding and antibonding orbitals.
Count the total number of electrons in bonding molecular orbitals (such as , , ).
Count the total number of electrons in antibonding molecular orbitals (such as , , ).
Plug these values into the bond order formula above.
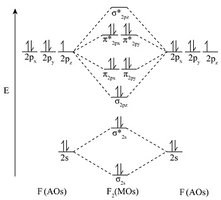
Try solving on your own before revealing the answer!
