 Back
BackGeneral Chemistry Study Guidance: Colligative Properties, Intermolecular Forces, and Phase Diagrams
Study Guide - Smart Notes

Q1. Benzene has a higher vapor pressure than toluene. Which compound has the higher boiling point?
Background
Topic: Vapor Pressure and Boiling Point
This question tests your understanding of the relationship between vapor pressure and boiling point for different substances.
Key Terms:
Vapor Pressure: The pressure exerted by a vapor in equilibrium with its liquid phase at a given temperature.
Boiling Point: The temperature at which the vapor pressure of a liquid equals the external pressure.
Step-by-Step Guidance
Recall that a higher vapor pressure means the liquid evaporates more easily at a given temperature.
Understand that the boiling point is reached when the vapor pressure equals atmospheric pressure.
Compare the vapor pressures: Benzene has a higher vapor pressure than toluene at the same temperature.
Think about which liquid needs to be heated more to reach atmospheric pressure.
Try solving on your own before revealing the answer!
Q2. If 10.0 grams of ethanol (molar mass = 46.0 g/mol) are mixed with 15 grams of water, what is the mole fraction of ethanol?
Background
Topic: Solution Concentration (Mole Fraction)
This question tests your ability to calculate mole fraction, a way of expressing concentration in a mixture.
Key Terms and Formulas:
Mole Fraction ():
Moles:
Step-by-Step Guidance
Calculate moles of ethanol:
Calculate moles of water:
Add the moles of ethanol and water to get the total moles.
Set up the mole fraction formula for ethanol.
Try solving on your own before revealing the answer!
Q3. Which of the following, when 10.0 g are dissolved in 1 liter of water, will result in the lowest freezing point for the solution?
Background
Topic: Colligative Properties (Freezing Point Depression)
This question tests your understanding of how solute particles affect the freezing point of a solution.
Key Terms and Formulas:
Freezing Point Depression:
= van't Hoff factor (number of particles the solute produces in solution)
= molality ()
Step-by-Step Guidance
Calculate the molality for each solute:
Determine the van't Hoff factor () for each solute based on how many ions or particles it produces.
Multiply and for each solute to compare the effect on freezing point.
Identify which solute produces the most particles in solution.
Try solving on your own before revealing the answer!
Q4. What is a supercritical fluid?
Background
Topic: Phase Diagrams and Supercritical Fluids
This question tests your understanding of the properties and conditions of supercritical fluids.
Key Terms:
Supercritical Fluid: A state of matter above its critical temperature and pressure, where distinct liquid and gas phases do not exist.
Critical Point: The temperature and pressure at which the liquid and gas phases of a substance become indistinguishable.
Step-by-Step Guidance
Identify the critical temperature and pressure for the substance in question.
Describe the properties of a supercritical fluid (density, solvating ability, etc.).
Explain how supercritical fluids are used in industry or research.
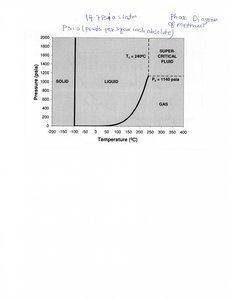
Try solving on your own before revealing the answer!
Q5. If the human eye has an osmotic pressure of 8.00 atm at 25°C, what molarity of solute particles in water will provide an isotonic eye drop solution?
Background
Topic: Osmosis and Colligative Properties
This question tests your ability to use the formula for osmotic pressure to calculate the required molarity for an isotonic solution.
Key Terms and Formulas:
Osmotic Pressure:
= osmotic pressure (atm)
= van't Hoff factor
= molarity (mol/L)
= gas constant ( L·atm/mol·K)
= temperature in Kelvin
Step-by-Step Guidance
Convert the temperature to Kelvin: K
Write the osmotic pressure equation:
Rearrange to solve for :
Plug in the values for , , and (and if known).
Try solving on your own before revealing the answer!
Q6. What volume of solution of sodium chloride with a density of 1.08 g/mL and a freezing point of -1.86°C contains 20.1 g of sodium chloride?
Background
Topic: Colligative Properties and Solution Density
This question tests your ability to relate mass, density, and freezing point depression to calculate the volume of a solution.
Key Terms and Formulas:
Freezing Point Depression:
Density:
Molality:
Step-by-Step Guidance
Calculate moles of sodium chloride:
Use freezing point depression to find the mass of solvent needed.
Calculate the total mass of solution (solute + solvent).
Use density to convert mass of solution to volume.
Try solving on your own before revealing the answer!
Q7. Why does molecule A have the higher boiling point than molecule B?
Background
Topic: Intermolecular Forces and Boiling Point
This question tests your understanding of how different types of intermolecular forces affect boiling points.
Key Terms:
London Dispersion Forces: Weak forces present in all molecules, stronger in larger or more polarizable molecules.
Dipole-Dipole Interactions: Forces between polar molecules.
Hydrogen Bonding: Strong dipole-dipole interaction involving H bonded to N, O, or F.
Step-by-Step Guidance
Examine the molecular structures for possible hydrogen bonding or dipole-dipole interactions.
Compare the strength of intermolecular forces present in each molecule.
Recall that stronger intermolecular forces lead to higher boiling points.
Try solving on your own before revealing the answer!
Q8. Between these two arrangements of a molecule, which has stronger intermolecular forces?
Background
Topic: Molecular Arrangement and Intermolecular Forces
This question tests your ability to compare the strength of intermolecular forces based on molecular arrangement.
Key Terms:
London Dispersion Forces: Increase with surface area and molecular contact.
Dipole-Dipole Interactions: Depend on molecular polarity and arrangement.
Step-by-Step Guidance
Analyze the arrangement of the molecules for surface area and contact.
Determine which arrangement allows for stronger intermolecular interactions.
Recall that more contact or greater surface area can increase the strength of London dispersion forces.
Try solving on your own before revealing the answer!
