 Back
BackGeneral Chemistry Study Guidance: Temperature, Significant Figures, Measurement, and Unit Conversions
Study Guide - Smart Notes

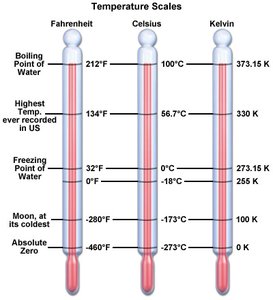
Q1. What are the three temperature scales represented in the model?
Background
Topic: Temperature Scales
This question is testing your understanding of the different temperature scales commonly used in chemistry and their relationships.
Key Terms:
Celsius (°C): Metric scale based on water's freezing and boiling points.
Fahrenheit (°F): Commonly used in the US, based on water's freezing and boiling points.
Kelvin (K): Absolute scale used in scientific calculations.
Step-by-Step Guidance
Examine the model and identify the three columns, each labeled with a different temperature scale.
Note the names and symbols for each scale.
Consider why each scale is important in chemistry, especially the Kelvin scale for gas law calculations.
Try solving on your own before revealing the answer!
Final Answer:
The three temperature scales are Fahrenheit (°F), Celsius (°C), and Kelvin (K).
These are the most commonly used scales in chemistry for measuring temperature.
Q2. What is the freezing point of water in the three temperature scales?
Background
Topic: Temperature Scale Reference Points
This question is testing your ability to recall the freezing point of water in different temperature scales, which is a fundamental reference in chemistry.
Key Terms:
Freezing Point: The temperature at which water changes from liquid to solid.

Step-by-Step Guidance
Look at the model and locate the freezing point of water for each scale.
Record the values for Fahrenheit, Celsius, and Kelvin.
Compare how the freezing point is represented differently in each scale.
Try solving on your own before revealing the answer!
Final Answer:
Freezing point of water: 32°F (Fahrenheit), 0°C (Celsius), 273.15 K (Kelvin).
These values are standard reference points for each scale.
Q3. What is the temperature of absolute zero for the 3 temperature scales? What does absolute zero represent?
Background
Topic: Absolute Zero and Temperature Scales
This question is testing your understanding of absolute zero, the lowest possible temperature, and its significance in chemistry.
Key Terms:
Absolute Zero: The point at which all thermal motion ceases.
Kelvin Scale: Starts at absolute zero.

Step-by-Step Guidance
Find the absolute zero value for each scale in the model.
Note the values for Fahrenheit, Celsius, and Kelvin.
Consider what absolute zero means physically (no thermal energy).
Try solving on your own before revealing the answer!
Final Answer:
Absolute zero: -460°F (Fahrenheit), -273°C (Celsius), 0 K (Kelvin).
Absolute zero represents the point where all molecular motion stops.
Q4. Which temperature scale can never produce a reading below zero?
Background
Topic: Absolute Temperature Scales
This question is testing your understanding of the properties of different temperature scales, especially the concept of absolute zero.
Key Terms:
Absolute Scale: A scale that starts at zero and cannot have negative values.
Kelvin Scale: Used in scientific calculations, always positive.

Step-by-Step Guidance
Review the model and note which scale starts at zero and does not go negative.
Recall why this scale is important for gas law calculations.
Compare with the other scales that allow negative values.
Try solving on your own before revealing the answer!
Final Answer:
The Kelvin scale can never produce a reading below zero.
This is because it is an absolute scale, starting at absolute zero.
Q5. The record high for DFW is 113 °F. Determine this temperature in Celsius and Kelvin.
Background
Topic: Temperature Conversion
This question is testing your ability to convert between Fahrenheit, Celsius, and Kelvin using mathematical relationships.
Key formulas:
(Fahrenheit to Celsius)
(Celsius to Kelvin)
Step-by-Step Guidance
Start with the given temperature: °F.
Use the formula to convert to Celsius.
Calculate first, then multiply by .
Once you have , use to convert to Kelvin.
Try solving on your own before revealing the answer!
Final Answer:
113 °F is approximately 45 °C and 318 K.
These conversions use the standard formulas for temperature conversion.
Q6. Examine the thermometer carefully. What is the temperature difference between the closest spaced increments/ticks on the thermometer for °C?
Background
Topic: Measurement Precision
This question is testing your ability to read a thermometer and understand the precision of its scale.
Key Terms:
Increment: The smallest division on the scale.
Precision: The degree to which repeated measurements under unchanged conditions show the same results.

Step-by-Step Guidance
Look at the thermometer image and identify the labeled values for °C.
Count the number of ticks between two labeled values (e.g., between 100°C and 150°C).
Divide the difference between labeled values by the number of ticks to find the increment.
Try solving on your own before revealing the answer!
Final Answer:
The temperature difference between the closest spaced increments is 10°C.
This is determined by counting the ticks and dividing the labeled difference.
Q7. In the four measurements made by the students, which “places” always have the same value?
Background
Topic: Measurement Consistency
This question is testing your understanding of significant digits and how certain digits remain consistent across repeated measurements.
Key Terms:
Certain Digits: Digits determined directly from the instrument's markings.
Estimated Digit: The last digit, which is a best guess.

Step-by-Step Guidance
Review the four measurements: 152°C, 151°C, 152°C, 153°C.
Identify which digits (hundreds, tens, ones) are consistent across all measurements.
Consider why these digits are consistent based on the thermometer's scale.
Try solving on your own before revealing the answer!
Final Answer:
The hundreds and tens places always have the same value in these measurements.
This is because the thermometer's scale allows for certainty in these digits.
Q8. Why is it easy for students to be in agreement on the values in the 10’s and 100’s places?
Background
Topic: Measurement Precision and Agreement
This question is testing your understanding of why certain digits are more reliable and consistent in measurements.
Key Terms:
Precision: Consistency of repeated measurements.
Instrument Markings: The scale divisions that allow for certainty in measurement.

Step-by-Step Guidance
Consider how the thermometer is marked and what digits are directly read from the scale.
Think about how the certainty of these digits leads to agreement among students.
Reflect on the role of estimated digits versus certain digits.
Try solving on your own before revealing the answer!
Final Answer:
It is easy for students to agree on the 10’s and 100’s places because these are directly read from the instrument's markings.
These digits are certain and not subject to estimation.
Q9. In the four measurements made by the students on this thermometer, which “place” commonly has some variability (or uncertainty)? Why does this “place” have some variability?
Background
Topic: Measurement Uncertainty
This question is testing your understanding of estimated digits and why variability occurs in measurements.
Key Terms:
Estimated Digit: The last digit in a measurement, representing uncertainty.
Measurement Variability: Differences due to estimation.

Step-by-Step Guidance
Review the four measurements and identify which digit varies.
Understand that this digit is estimated, not directly read from the scale.
Consider why estimation leads to variability among different students.
Try solving on your own before revealing the answer!
Final Answer:
The ones place commonly has variability because it is the estimated digit.
This digit is subject to interpretation and estimation, leading to differences.
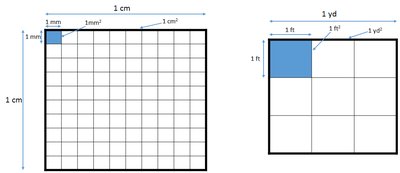
Q10. Let’s consider length. How many millimeters (mm) are in 1 centimeter (cm)? Write this as an equality.
Background
Topic: Metric Unit Conversions
This question is testing your ability to convert between metric units using SI prefixes.
Key Terms:
Centimeter (cm): 1/100 of a meter.
Millimeter (mm): 1/1000 of a meter.
Step-by-Step Guidance
Recall the relationship between centimeters and millimeters from the SI prefix table.
Use the model to visualize how many millimeters fit into one centimeter.
Write the equality based on the conversion factor.
Try solving on your own before revealing the answer!
Final Answer:
1 cm = 10 mm.
This is a standard metric conversion based on SI prefixes.
Q11. Now, consider an area that is 1 centimeter by 1 centimeter as shown above. Explain how the numerical value of 1 was obtained and the unit of cm².
Background
Topic: Area Units in Metric System
This question is testing your understanding of how area is calculated and represented in metric units.
Key Terms:
Area: The amount of space within a boundary, measured in square units.
cm²: Square centimeters, the unit for area.

Step-by-Step Guidance
Visualize a square with sides of 1 cm each.
Calculate the area: .
Understand that the unit cm² represents the area of a square with 1 cm sides.
Try solving on your own before revealing the answer!
Final Answer:
The area is 1 cm² because it is a square with sides of 1 cm.
Area is calculated by multiplying the length and width.
Q12. In the model, 1 mm² is illustrated (shaded in blue). How many mm² are in 1 cm²? Write this as an equality.
Background
Topic: Area Unit Conversions
This question is testing your ability to convert between square units in the metric system.
Key Terms:
mm²: Square millimeters.
cm²: Square centimeters.

Step-by-Step Guidance
Recall that 1 cm = 10 mm.
Calculate the area: .
Write the equality based on this calculation.
Try solving on your own before revealing the answer!
Final Answer:
1 cm² = 100 mm².
This is because each side of the square is 10 mm, so the area is 10 × 10 = 100 mm².
Q13. Refer to information in the right of the model showing the relationship between feet (ft) and yards (yd). How many feet are in 1 yard (write as an equality)?
Background
Topic: Imperial Unit Conversions
This question is testing your ability to convert between feet and yards, which is important for dimensional analysis.
Key Terms:
Foot (ft): Imperial unit of length.
Yard (yd): Imperial unit of length.

Step-by-Step Guidance
Recall the standard conversion: 1 yard = 3 feet.
Write the equality based on this relationship.
Visualize the model to reinforce the conversion.
Try solving on your own before revealing the answer!
Final Answer:
1 yard = 3 feet.
This is a standard conversion in the imperial system.
Q14. Now, consider an area that is 1 yard by 1 yard. How many ft² are in 1 yd²? Write this as an equality.
Background
Topic: Area Unit Conversions (Imperial)
This question is testing your ability to convert between square units in the imperial system.
Key Terms:
ft²: Square feet.
yd²: Square yards.

Step-by-Step Guidance
Recall that 1 yard = 3 feet.
Calculate the area: .
Write the equality based on this calculation.
Try solving on your own before revealing the answer!
Final Answer:
1 yd² = 9 ft².
This is because each side of the square is 3 ft, so the area is 3 × 3 = 9 ft².
