 Back
BackGeneral Chemistry Study Guide: Atomic Structure, Electron Configuration, and Light
Study Guide - Smart Notes

Q1. Calculate the wavelength or frequency of electromagnetic radiation given one or the other.
Background
Topic: Electromagnetic Radiation and Light
This question tests your understanding of the relationship between wavelength, frequency, and the speed of light. These concepts are foundational in atomic structure and quantum chemistry.
Key Terms and Formulas
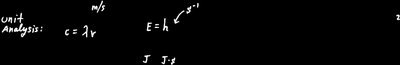
Wavelength (): The distance between two consecutive peaks of a wave (measured in meters).
Frequency (): The number of wave cycles that pass a point per second (measured in s or Hz).
Speed of light (): m/s (in a vacuum).
Key formula:
Step-by-Step Guidance
Identify what is given in the problem (either wavelength or frequency) and what you need to find.
Write down the relationship between the speed of light, wavelength, and frequency: .
Rearrange the equation to solve for the unknown variable. For example, if you are given frequency and need to find wavelength: .
Make sure your units are consistent (wavelength in meters, frequency in Hz, speed of light in m/s).
Try solving on your own before revealing the answer!
Q2. Calculate the energy of a photon given its frequency or wavelength.
Background
Topic: Quantum Theory of Light
This question tests your ability to use Planck's equation to relate the energy of a photon to its frequency or wavelength.
Key Terms and Formulas
Planck's constant (): J·s
Energy of a photon ():
Alternatively, using wavelength:

Step-by-Step Guidance
Determine whether you are given frequency () or wavelength ().
If given frequency, use .
If given wavelength, use .
Plug in the values for Planck's constant, speed of light, and the given frequency or wavelength (make sure units are correct: in meters, in Hz).
Try solving on your own before revealing the answer!
Q3. Write the electron configuration for a given element and determine the number of valence electrons.
Background
Topic: Electron Configuration and Periodic Table
This question tests your understanding of how electrons are arranged in atoms and how to use the periodic table to determine electron configurations and valence electrons.
Key Terms and Formulas
Electron configuration: The arrangement of electrons in an atom's orbitals.
Valence electrons: Electrons in the outermost shell, important for chemical bonding.

Step-by-Step Guidance
Find the atomic number of the element to determine the total number of electrons.
Fill the electrons into orbitals following the Aufbau principle (1s, 2s, 2p, 3s, 3p, etc.).
Write out the full electron configuration or use the noble gas shorthand if appropriate.
Identify the electrons in the outermost shell (highest principal quantum number) to determine the number of valence electrons.
Try solving on your own before revealing the answer!
Q4. Use Lewis symbols to represent atoms and predict the number of bonds an atom can form.
Background
Topic: Lewis Structures and Chemical Bonding
This question tests your ability to use Lewis symbols to represent valence electrons and predict bonding patterns for main group elements.
Key Terms and Formulas
Lewis symbol: A representation of an atom showing its valence electrons as dots around the element symbol.
Octet rule: Atoms tend to gain, lose, or share electrons to achieve a full set of eight valence electrons.

Step-by-Step Guidance
Determine the number of valence electrons for the atom using its group number or electron configuration.
Draw the Lewis symbol by placing dots around the element symbol to represent valence electrons.
Predict how many electrons the atom needs to gain, lose, or share to achieve an octet (or duet for hydrogen and helium).
Use this information to predict the number of bonds the atom can form.
Try solving on your own before revealing the answer!
