 Back
BackGeneral Chemistry Study Guide: Atoms, Elements, Molecules, and Compounds
Study Guide - Smart Notes

Atoms & Elements
Atomic Structure and Isotopes
Atoms are the fundamental units of matter, composed of protons, neutrons, and electrons. Isotopes are variants of elements with the same number of protons but different numbers of neutrons.
Protons: Positively charged particles found in the nucleus; the atomic number equals the number of protons.
Neutrons: Neutral particles in the nucleus; isotopes differ in neutron count.
Electrons: Negatively charged particles orbiting the nucleus; in a neutral atom, electrons equal protons.
Isotope Example: 200Hg has 80 protons and 120 neutrons.
Atomic Mass Calculation: Weighted average based on isotope abundance and mass.
Formula:
Periodic Table Organization
The periodic table arranges elements by increasing atomic number and groups them by similar properties.
Main Group Elements: Groups 1A–8A, including alkali metals, alkaline earth metals, halogens, and noble gases.
Transition Metals: Elements in the center of the table, often forming multiple ions.
Metalloids: Elements with properties intermediate between metals and nonmetals.
Molecules and Compounds
Naming Ionic and Molecular Compounds
Compounds are named according to their composition and the rules for ionic or molecular compounds.
Ionic Compounds: Formed from metals and nonmetals; names include the cation and anion, with Roman numerals for variable charge metals (e.g., tin(IV) sulfate for Sn(SO4)2).
Molecular Compounds: Formed from nonmetals; use prefixes to indicate the number of atoms (e.g., diphosphorus tetrafluoride for P2F4).
Hydrates: Compounds with water molecules attached; indicated by prefixes (e.g., octahydrate).
Polyatomic Ions
Polyatomic ions are charged groups of covalently bonded atoms. They are common in many compounds.
Examples: Sulfate (SO42–), Nitrate (NO3–), Ammonium (NH4+).
Importance: Polyatomic ions are used in naming and writing formulas for compounds.
Empirical and Molecular Formulas
The empirical formula shows the simplest ratio of elements in a compound, while the molecular formula shows the actual number of atoms.
Empirical Formula: Determined from percent composition.
Molecular Formula: Determined from molar mass and empirical formula.
Formula:
Mathematical Operations and Functions
Mole Calculations
The mole is a fundamental unit for counting atoms and molecules. Calculations often involve converting between mass, moles, and number of particles.
Mole Formula:
Avogadro’s Number: particles per mole.
Number of Molecules:
Mass Percent and Molar Mass
Mass percent expresses the proportion of each element in a compound. Molar mass is the mass of one mole of a substance.
Mass Percent Formula:
Molar Mass: Sum of atomic masses of all atoms in the formula.
Bonus: Lab Techniques and Procedures
Hydrates and Formula Writing
Hydrates are compounds with water molecules attached. The formula includes a dot and the number of water molecules (e.g., CaCl2·6H2O).
Writing Hydrate Formulas: Use the compound formula followed by a dot and the number of water molecules.
Example: Chromium(III) sulfite octahydrate is written as Cr2(SO3)3·8H2O.
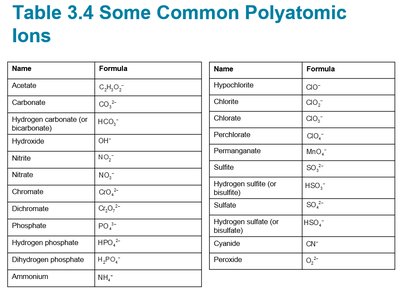
Summary Table: Common Polyatomic Ions
The following table summarizes common polyatomic ions used in naming and writing chemical formulas:
Name | Formula |
|---|---|
Acetate | C2H3O2– |
Carbonate | CO32– |
Hydrogen carbonate (bicarbonate) | HCO3– |
Hydroxide | OH– |
Nitrate | NO3– |
Chromate | CrO42– |
Dichromate | Cr2O72– |
Phosphate | PO43– |
Hydrogen phosphate | HPO42– |
Dihydrogen phosphate | H2PO4– |
Ammonium | NH4+ |
Hypochlorite | ClO– |
Chlorite | ClO2– |
Chlorate | ClO3– |
Perchlorate | ClO4– |
Permanganate | MnO4– |
Sulfite | SO32– |
Hydrogen sulfite (bisulfite) | HSO3– |
Sulfate | SO42– |
Hydrogen sulfate (bisulfate) | HSO4– |
Cyanide | CN– |
Peroxide | O22– |
