 Back
BackGeneral Chemistry Study Guide: Chapters 1 & 2
Study Guide - Smart Notes

Classification of Matter
Types of Matter
The classification of matter is fundamental in chemistry, distinguishing substances based on their composition and properties.
Pure Substances: Matter with a fixed composition. Includes elements (e.g., O2, Fe) and compounds (e.g., H2O, NaCl).
Mixtures: Physical combinations of two or more substances. Can be homogeneous (uniform, e.g., saltwater) or heterogeneous (non-uniform, e.g., sand and water).
States of Matter: Solid (fixed shape and volume), liquid (fixed volume, variable shape), gas (variable shape and volume).
Elements vs. Compounds vs. Mixtures: Elements are pure substances of one type of atom; compounds are pure substances of two or more atoms chemically bonded; mixtures are physical blends.
Atomic Symbols: Each element is represented by a unique symbol (e.g., H for hydrogen, O for oxygen).
Chemical and Physical Changes
Understanding the difference between chemical and physical changes is essential for identifying reactions and processes.
Physical Change: Alters the form or appearance but not the composition (e.g., melting ice).
Chemical Change: Produces new substances with different properties (e.g., burning wood).
Indicators of Chemical Change: Color change, gas production, formation of a precipitate, energy change.
Units of Measurement
SI Units and Prefixes
Chemistry relies on standardized units for measurement. Prefixes modify base units to represent larger or smaller quantities.
Common Prefixes: Kilo (k, ), Milli (m, ), Giga (G, ), etc.
Examples: 1 kilometer (km) = 1000 meters (m); 1 milligram (mg) = 0.001 grams (g).
Temperature Conversions
Temperature is measured in Celsius (°C), Fahrenheit (°F), and Kelvin (K). Conversion formulas are essential for calculations.
Conversion Equations:
Significant Figures
Rules and Application
Significant figures reflect the precision of measurements and calculations in chemistry.
Rules: All nonzero digits are significant; zeros between nonzero digits are significant; leading zeros are not significant; trailing zeros are significant if there is a decimal point.
Determining Significant Figures: Count digits according to the rules above.
Accuracy vs. Precision: Accuracy is how close a measurement is to the true value; precision is how reproducible measurements are.
Density
Definition and Calculation
Density is a physical property defined as mass per unit volume.
Formula:
Units: Commonly g/cm3 or kg/m3.
Application: Used to identify substances and solve problems involving mass and volume.
Dimensional Analysis
Unit Conversion
Dimensional analysis uses conversion factors to change units in calculations.
Conversion Factors: Equalities such as 1 mile = 1609.34 meters, 1 inch = 2.54 cm.
Example: To convert miles to kilometers, multiply by the conversion factor.
Atomic Theory and Structure
Historical Models
The atomic theory explains the nature of matter and the structure of atoms.
Dalton's Postulates: Matter is composed of atoms; atoms of the same element are identical; atoms combine in simple ratios to form compounds.
Rutherford Model: Gold foil experiment showed that atoms have a small, dense nucleus.
Atomic Structure
Atoms consist of a nucleus (protons and neutrons) surrounded by electrons.
Protons: Positive charge (+1), found in nucleus.
Neutrons: No charge, found in nucleus.
Electrons: Negative charge (-1), orbit nucleus.
Atomic Symbols: Show element, mass number, and atomic number.
Isotopes: Atoms of the same element with different numbers of neutrons.
Atomic Number (Z): Number of protons.
Mass Number (A): Number of protons + neutrons.
Atomic Weight: Weighted average of isotopic masses.
Atomic Weights and Isotopes
Isotope Identification and Calculations
Isotopes are variants of elements with different neutron counts. Atomic weight is calculated from isotopic abundances.
Isotope: Same element, different mass number.
Average Atomic Mass Formula:
Application: Calculate average atomic mass using isotope masses and fractional abundances.
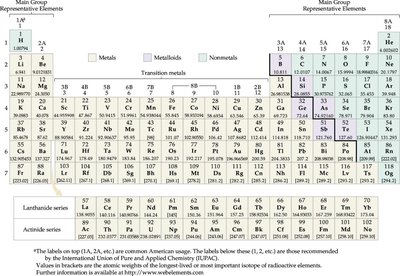
Periodic Table
Structure and Properties
The periodic table organizes elements by atomic number and properties. It is divided into groups (columns) and periods (rows).
Groups: Vertical columns; elements in a group share similar properties.
Periods: Horizontal rows; properties change progressively across a period.
Metals: Left side; conduct electricity, malleable, shiny.
Nonmetals: Right side; poor conductors, brittle.
Metalloids: Border between metals and nonmetals; intermediate properties.
Noble Gases: Group 18; inert, stable.
Element Properties: Can be inferred from location in the table.
Molecules and Compounds
Formulas and Naming
Molecules are composed of atoms bonded together. Compounds can be represented by molecular and empirical formulas.
Molecular Formula: Shows actual number of atoms in a molecule (e.g., C6H12O6).
Empirical Formula: Shows simplest ratio of atoms (e.g., CH2O for glucose).
Naming Molecular Compounds: Use prefixes to indicate number of atoms (e.g., carbon dioxide, CO2).
Ions and Ionic Compounds
Formation and Naming
Ions are atoms or molecules with a net charge. Ionic compounds are formed from cations and anions.
Cations: Positive ions (e.g., Na+).
Anions: Negative ions (e.g., Cl-).
Predicting Ionic Charges: Based on group number in periodic table.
Formulas for Ionic Compounds: Combine ions to achieve charge neutrality (e.g., Na+ + Cl- → NaCl).
Molecular vs. Ionic Compounds: Molecular compounds have covalent bonds; ionic compounds have electrostatic attraction.
Naming Ionic Compounds
Names are derived from the cation and anion. For example, NaCl is sodium chloride.
Rules: Name cation first, then anion (with -ide ending).
Periodic Table Data Table
Protons, Neutrons, Electrons, Net Charge, Mass Number
Use the periodic table to complete a table of atomic properties for elements and ions.
Element/Ion | Protons | Neutrons | Electrons | Net Charge | Mass Number |
|---|---|---|---|---|---|
Na | 11 | 12 | 11 | 0 | 23 |
Na+ | 11 | 12 | 10 | +1 | 23 |
Cl | 17 | 18 | 17 | 0 | 35 |
Cl- | 17 | 18 | 18 | -1 | 35 |
O | 8 | 8 | 8 | 0 | 16 |
O2- | 8 | 8 | 10 | -2 | 16 |
Additional info: Table entries can be completed for any element or ion using periodic table data. |
