 Back
BackGeneral Chemistry Study Guide: Chapters 1–3
Study Guide - Smart Notes

Mathematical Operations and Functions
Addition, Subtraction, Multiplication, and Division
Mathematical operations are fundamental in chemistry for solving quantitative problems. Addition and subtraction are used to combine or compare values, while multiplication and division are essential for calculations involving ratios, rates, and conversions.
Addition/Subtraction: Used for combining measurements or calculating differences.
Multiplication/Division: Used for scaling values, calculating densities, and converting units.
Example: If you add 2.5 g and 3.1 g, the sum is 5.6 g.
Significant Figures and Scientific Notation
Significant figures reflect the precision of a measurement. Scientific notation is used to express very large or small numbers in a compact form.
Significant Figures: The digits in a measurement that are known with certainty plus one estimated digit.
Scientific Notation: Numbers are written as , where a is a number between 1 and 10 and n is an integer.
Example: 0.00045 can be written as .
Atoms & Elements
Element Symbols, Names, and Classification
Each element is represented by a unique symbol and can be classified as a metal, nonmetal, or metalloid. The periodic table organizes elements based on their properties.
Element Symbol: One or two letters representing an element (e.g., H for hydrogen).
Classification: Metals are typically shiny and conductive, nonmetals are dull and insulating, and metalloids have properties of both.
Example: Si (Silicon) is a metalloid.

Chemical Reactions
Classification of Matter
Matter can be classified as an element, compound, homogeneous mixture, or heterogeneous mixture.
Element: Pure substance consisting of one type of atom.
Compound: Substance formed from two or more elements chemically bonded.
Homogeneous Mixture: Uniform composition throughout (e.g., saltwater).
Heterogeneous Mixture: Non-uniform composition (e.g., sand and water).
Physical vs. Chemical Properties
Properties of matter are classified as physical or chemical.
Physical Property: Can be observed without changing the substance (e.g., melting point).
Chemical Property: Describes how a substance reacts (e.g., flammability).
Example: Boiling water is a physical change; rusting iron is a chemical change.
Chemical Quantities & Aqueous Reactions
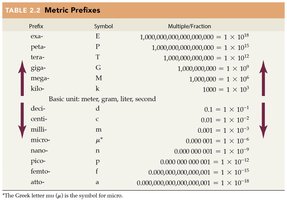
Unit Conversions and Metric Prefixes
Unit conversions are essential for expressing measurements in different units. Metric prefixes indicate multiples or fractions of base units.
Metric Prefixes: Prefixes such as kilo-, milli-, and micro- represent powers of ten.
Conversion Example: 1 kilometer (km) = 1,000 meters (m).
Prefix | Symbol | Multiple/Fraction |
|---|---|---|
kilo | K | 1,000 = |
milli | m | 0.001 = |
micro | \mu | 0.000001 = |
nano | n | 0.000000001 = |
pico | p | 0.000000000001 = |
mega | M | 1,000,000 = |
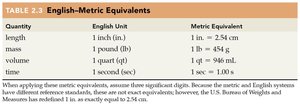
English-Metric Equivalents
Conversions between English and metric units are often required in laboratory and real-world settings.
Quantity | English Unit | Metric Equivalent |
|---|---|---|
Length | 1 inch (in.) | 2.54 cm |
Mass | 1 pound (lb) | 454 g |
Volume | 1 quart (qt) | 946 mL |
Time | 1 second (sec) | 1.00 s |
Density Calculations
Density is a physical property defined as mass per unit volume.
Formula:
Example: If a substance has a mass of 10 g and a volume of 2 mL, its density is .
The Percent Concept
Percentages are used to express ratios, concentrations, and yields in chemistry.
Formula:
Example: If 20 g of salt is dissolved in 100 g of water, the percent salt is .
Intro to General Chemistry
Changes of Physical State
Substances can change between solid, liquid, and gas states through physical processes.
Evaporation: Liquid to gas
Condensation: Gas to liquid
Melting: Solid to liquid
Freezing: Liquid to solid
Sublimation: Solid to gas
Deposition: Gas to solid
Law of Conservation of Mass
The law of conservation of mass states that mass is neither created nor destroyed in a chemical reaction.
Formula:
Example: If 10 g of reactants produce 10 g of products, mass is conserved.
Law of Conservation of Energy
The law of conservation of energy states that energy cannot be created or destroyed, only transformed.
Example: Chemical energy in fuel is converted to heat and light during combustion.
Additional info: Academic context and explanations have been expanded for clarity and completeness. Tables have been recreated and images included only where directly relevant to the explanation.
