 Back
BackGeneral Chemistry Study Guide: Electrolytes, Solubility, Precipitation, Molarity, Stoichiometry, and Limiting Reactants
Study Guide - Smart Notes

Q1. Classify each compound as a strong electrolyte, weak electrolyte, or none:
NaCl
CH₃COOH
HCl
NH₃
C₆H₁₂O₆ (glucose)
Ba(OH)₂
HF
Background
Topic: Electrolytes in Solution
This question tests your understanding of how substances behave in water—whether they dissociate completely, partially, or not at all, and how that affects their classification as strong, weak, or non-electrolytes.
Key Terms:
Electrolyte: A substance that produces ions when dissolved in water.
Strong Electrolyte: Completely dissociates into ions.
Weak Electrolyte: Partially dissociates into ions.
Non-Electrolyte: Does not dissociate into ions.
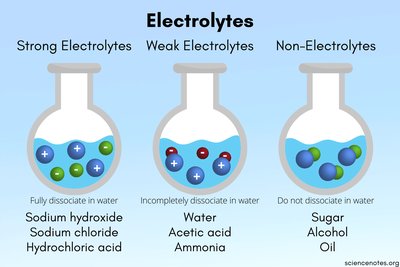
Step-by-Step Guidance
Recall that ionic compounds (like NaCl) and strong acids/bases (like HCl, Ba(OH)₂) are typically strong electrolytes because they fully dissociate in water.
Weak acids (like CH₃COOH) and weak bases (like NH₃) only partially dissociate, making them weak electrolytes.
Covalent compounds (like glucose, C₆H₁₂O₆) do not dissociate into ions and are non-electrolytes.
For HF, consider its behavior: it is a weak acid and only partially dissociates.
Use the image above to reinforce your understanding of how each type behaves in water.
Try classifying each compound before checking the answer!
Final Answer:
NaCl: Strong electrolyte
CH₃COOH: Weak electrolyte
HCl: Strong electrolyte
NH₃: Weak electrolyte
C₆H₁₂O₆ (glucose): None (non-electrolyte)
Ba(OH)₂: Strong electrolyte
HF: Weak electrolyte
Strong electrolytes fully dissociate, weak electrolytes partially dissociate, and non-electrolytes do not dissociate in water.
Q2. Predict if each compound is soluble or insoluble in water:
AgCl
Na₂SO₄
BaCO₃
KNO₃
PbI₂
(NH₄)₃PO₄
Background
Topic: Solubility Rules for Ionic Compounds
This question tests your ability to use solubility rules to determine whether a compound will dissolve in water or form a precipitate.
Key Terms and Solubility Rules:
Soluble: Compound dissolves in water.
Insoluble: Compound does not dissolve in water.
Refer to the solubility rules table for guidance.
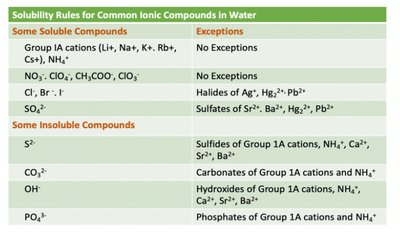
Step-by-Step Guidance
For each compound, identify the cation and anion.
Check the solubility rules table for the general rule and any exceptions.
For example, AgCl contains Cl⁻ (usually soluble), but Ag⁺ is an exception (insoluble).
Repeat this process for each compound, considering both the general rule and exceptions.
Use the table to reinforce your reasoning for each prediction.
Try predicting solubility for each compound before checking the answer!
Final Answer:
AgCl: Insoluble
Na₂SO₄: Soluble
BaCO₃: Insoluble
KNO₃: Soluble
PbI₂: Insoluble
(NH₄)₃PO₄: Soluble
Solubility rules help predict whether a compound will dissolve in water or form a precipitate.
