 Back
BackGeneral Chemistry Study Guide: Mass Relationships and Reactions in Aqueous Solution
Study Guide - Smart Notes

Mass Relationships in Chemical Reactions
Stoichiometry and Chemical Equations
Stoichiometry is the quantitative study of reactants and products in a chemical reaction. Balanced chemical equations provide the identities and relative amounts of substances involved, allowing chemists to predict the outcomes of reactions.
Balanced Chemical Equation: Shows the reactants and products with their correct coefficients.
Stoichiometric Ratio: The ratio of moles of reactants and products as indicated by the equation.
Steps to Balance Equations:
Translate the statement into a skeleton equation.
Balance atoms on both sides of the equation.
Adjust coefficients to the smallest whole numbers.
Check that the equation is balanced.
Specify the physical states of each substance.
Mass, Moles, and Molecules
Chemists use the concept of the mole to relate mass, number of particles, and chemical formulas. The mole is a counting unit that allows conversion between mass, moles, and number of molecules using Avogadro's number and molar mass.
Molar Mass (g/mol): The mass of one mole of a substance.
Avogadro's Number: entities per mole.
Conversion Steps: Mass → Moles → Molecules (or atoms).
Empirical Formula Determination
The empirical formula represents the simplest whole-number ratio of elements in a compound. It is determined by converting masses of elements to moles and finding the lowest whole-number ratio.
Steps:
Find mass of each element.
Convert mass to moles using molar mass.
Divide by the lowest number of moles to get the ratio.
Convert ratio to lowest whole numbers for empirical formula.
Theoretical Yield, Limiting Reactant, and Percent Yield
Theoretical yield is the maximum amount of product that can be formed from given reactants. The limiting reactant is the reactant that is completely consumed first, limiting the amount of product formed. Percent yield compares actual yield to theoretical yield.
Theoretical Yield: Calculated from the limiting reactant.
Percent Yield Formula:
Reactions in Aqueous Solution
Types of Chemical Reactions
Chemical reactions in aqueous solution can be classified into several types based on the patterns of reactant and product formation.
Combination (Synthesis): Two or more substances combine to form one product.
Decomposition: One substance breaks down into two or more products.
Single Displacement: One element replaces another in a compound.
Double Displacement: Exchange of ions between two compounds.
Combustion: A substance reacts with oxygen to produce energy, CO2, and H2O.
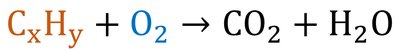
Electrolytes and Solution Chemistry
Electrolytes are substances that dissociate into ions when dissolved in water. Strong electrolytes dissociate completely, while weak electrolytes dissociate partially. Ionic compounds separate into ions in solution, whereas molecular compounds do not.
Strong Electrolytes: Complete dissociation into ions.
Weak Electrolytes: Partial dissociation into ions.
Molarity Formula:
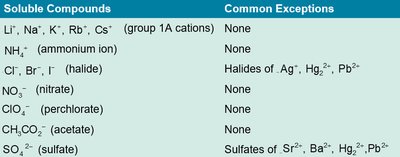
Solubility Rules
Solubility rules help predict whether a compound will dissolve in water. Some compounds are always soluble, while others have common exceptions.
Soluble Compounds | Common Exceptions |
|---|---|
Li+, Na+, K+, Rb+, Cs+ (group 1A cations) | None |
NH4+ (ammonium ion) | None |
Cl-, Br-, I- (halide) | Halides of Ag+, Hg22+, Pb2+ |
NO3- (nitrate) | None |
ClO4- (perchlorate) | None |
CH3CO2- (acetate) | None |
SO42- (sulfate) | Sulfates of Sr2+, Ba2+, Hg22+, Pb2+ |
Insoluble Compounds | Common Exceptions |
|---|---|
CO32- (carbonate) | Carbonates of group 1A cations, NH4+ |
S2- (sulfide) | Sulfides of group 1A cations, NH4+, Ca2+, Sr2+, Ba2+ |
PO43- (phosphate) | Phosphates of group 1A cations, NH4+ |
OH- (hydroxide) | Hydroxides of group 1A cations, Ca2+, Sr2+, Ba2+ |

Types of Aqueous Reactions
Common reactions in aqueous solution include precipitation, acid-base neutralization, and oxidation-reduction (redox) reactions.
Precipitation: Formation of an insoluble product (precipitate).
Acid-Base Neutralization: Acid reacts with base to form water and salt.
Oxidation-Reduction (Redox): Involves transfer of electrons between species.
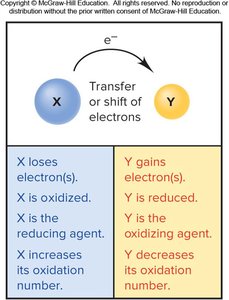
Oxidation-Reduction (Redox) Reactions
Redox Concepts and Oxidation Numbers
Redox reactions involve the transfer of electrons. Oxidation is the loss of electrons, and reduction is the gain of electrons. The oxidizing agent is reduced, and the reducing agent is oxidized. Oxidation numbers help track electron transfer.
Oxidation Number (O.N.): The hypothetical charge an atom would have if all bonds were ionic.
Rules:
Elemental form: O.N. = 0
Monatomic ion: O.N. = ion charge
Sum of O.N. in molecule = 0; in polyatomic ion = ion charge
Group 1: O.N. = +1; Group 2: O.N. = +2
Hydrogen: O.N. = +1 (with nonmetals), -1 (with metals)
Oxygen: O.N. = -2 (except peroxides: -1)
Group 17: O.N. = -1 (with metals/nonmetals except O)
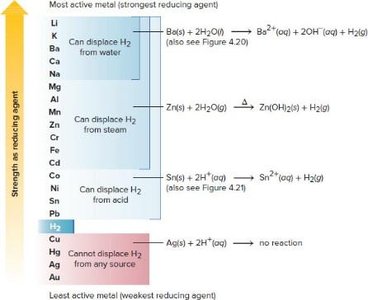
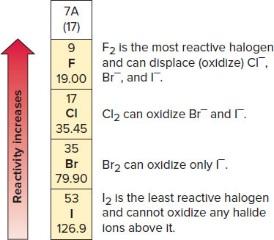
Activity Series and Halogen Reactivity
The activity series ranks metals and halogens by their ability to act as reducing or oxidizing agents. More active metals can displace less active metals from compounds, and more reactive halogens can oxidize less reactive halide ions.
Activity Series: Used to predict whether a reaction will occur.
Halogen Reactivity: F2 is most reactive, I2 is least.
Example: Combustion Analysis
Combustion analysis is used to determine the empirical formula of a compound by measuring the masses of CO2 and H2O produced.
Procedure: Burn a known mass of compound, measure products, calculate moles of C and H, determine empirical formula.
 Additional info: Academic context and explanations have been expanded for clarity and completeness.
Additional info: Academic context and explanations have been expanded for clarity and completeness.
