 Back
BackGeneral Chemistry Study Notes: Foundations, Measurements, Matter, and Atoms
Study Guide - Smart Notes

Chemistry in Our Lives
Scientific Method
The scientific method is a systematic approach used by scientists to investigate phenomena, acquire new knowledge, or correct and integrate previous knowledge. It involves several key steps:
Observation: Gathering information by measuring events in nature.
Hypothesis: A testable statement that explains the observations. It is modified if found incorrect.
Experiment: Procedures designed to test the hypothesis.
Theory: A model that describes how observations occur, supported by experimental results.
Example: If a blender doesn’t turn on, the observation is the malfunction, the hypothesis might be a broken motor, the experiment could be plugging it into another outlet, and the theory is that the blender needs repair.
Branches of Chemistry
Chemistry is divided into several branches, each focusing on different aspects:
General Chemistry: Basic principles and concepts.
Organic Chemistry: Study of carbon-containing compounds.
Biochemistry: Chemistry of living organisms.
Geochemistry: Chemistry of Earth materials.
Physical Chemistry: Interactions between energy and matter.
What Is Chemistry?
Chemistry is the study of substances in terms of their composition, structure, properties, and reactions. All matter, including water, food, and air, is composed of chemicals.
Chemicals
A chemical is a substance with a constant composition and properties. Examples include toothpaste, soap, food additives, plastics, and metals.
Learning Chemistry Effectively
Read before lectures.
Practice sample problems before checking answers.
Use study groups and ask questions during office hours.
Practice consistently; avoid cramming.
Mathematical Operations and Functions
Key Math Skills for Chemistry
Place Values: Understanding the value of digits in numbers (e.g., 2518: 2 thousands, 5 hundreds, etc.).
Positive & Negative Numbers: Rules for multiplication, division, addition, and subtraction.
Percentages: Percent = (part \div whole) \times 100. Example: 325 mg aspirin in 545 mg tablet: % aspirin = (325 \div 545) \times 100.
Solving Equations: Isolate the variable by performing the same operation on both sides. Example: 2x + 8 = 14, x = 3.
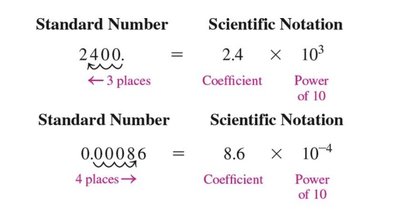
Scientific Notation
Scientific notation is used to express very large or very small numbers. The format is coefficient \times 10^n, where the coefficient is at least 1 but less than 10.
Positive exponent: Move decimal to the right.
Negative exponent: Move decimal to the left.
Examples:
3500 = 3.5 \times 10^3
0.000016 = 1.6 \times 10^{-5}
Measurements and Units
International System of Units (SI)
The SI system is the standard for scientific measurements. Key quantities and their units:
Quantity | Metric Unit | SI Unit | Abbreviation |
|---|---|---|---|
Length | meter | meter | m |
Volume | liter | cubic meter | L, m³ |
Mass | gram | kilogram | g, kg |
Temperature | Celsius | kelvin | °C, K |
Time | second | second | s |
Metric Prefixes
Kilo (k): 10^3
Hecto (h): 10^2
Deka (da): 10^1
Deci (d): 10^{-1}
Centi (c): 10^{-2}
Milli (m): 10^{-3}
Micro (µ): 10^{-6}
Key Metric Equalities
1 m = 100 cm = 1000 mm
1 L = 1000 mL
1 kg = 1000 g
Measured vs Exact Numbers
Measured Numbers: Obtained using measuring tools; have uncertainty and significant figures.
Exact Numbers: Counting items or defined conversions; unlimited significant figures.
Significant Figures (SF)
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Leading zeros are not significant.
Trailing zeros with a decimal are significant.
Trailing zeros without a decimal are not significant.
Density
Density is the mass per unit volume of a substance.
Formula:
Units: g/mL or g/cm³ (liquids & solids), g/L (gases)
Matter & Energy
Classification of Matter
Pure Substances: Fixed composition; includes elements and compounds.
Elements: One type of atom; cannot be broken down further.
Compounds: Two or more elements chemically combined in a definite ratio.
Mixtures: Two or more substances physically mixed; variable composition.
Homogeneous Mixtures: Uniform throughout (e.g., air, sugar water).
Heterogeneous Mixtures: Non-uniform; visibly different parts (e.g., salad, oil and water).
States and Properties of Matter
Solid: Definite shape and volume; particles vibrate in fixed positions.
Liquid: Definite volume, no definite shape; particles move slowly and randomly.
Gas: No definite shape or volume; particles move fast and are far apart.
Physical vs Chemical Properties and Changes
Physical Properties: Observed without changing identity (e.g., color, density).
Physical Changes: Substance remains the same; only appearance or state changes (e.g., melting ice).
Chemical Properties: Ability to form new substances (e.g., burning, rusting).
Chemical Changes: New substance is formed; chemical composition changes (e.g., burning a candle).
Temperature Scales and Conversions
Celsius (°C): Used in science.
Fahrenheit (°F): Common in the US.
Kelvin (K): SI unit.
Reference Points: Freezing water: 0 °C = 32 °F = 273 K; Boiling water: 100 °C = 212 °F = 373 K
Celsius to Fahrenheit:
Fahrenheit to Celsius:
Celsius to Kelvin:
Energy and Specific Heat
Energy: Ability to do work; includes kinetic (motion) and potential (stored) energy.
Heat Equation:
Where q = heat (J or cal), m = mass (g), SH = specific heat, ΔT = change in temperature (°C)
Atoms & Elements
Elements & Chemical Symbols
Element: Pure substance made of one type of atom.
Chemical Symbol: 1–2 letters; first capitalized, second lowercase (e.g., Co = cobalt).
Periodic Table Basics
Groups: Vertical columns; similar properties.
Periods: Horizontal rows.
Group Names: Alkali metals (1A), Alkaline earth metals (2A), Halogens (7A), Noble gases (8A).
Metals, Nonmetals & Metalloids
Metals: Shiny, malleable, ductile, good conductors, mostly solids.
Nonmetals: Dull, brittle, poor conductors, low melting points.
Metalloids: Semiconductors; properties intermediate between metals and nonmetals.
Structure of the Atom
Particle | Charge | Location | Mass |
|---|---|---|---|
Proton | +1 | Nucleus | ~1 amu |
Neutron | 0 | Nucleus | ~1 amu |
Electron | -1 | Outside nucleus | ~0 amu |
Atoms are neutral: protons = electrons.
Atomic Number & Mass Number
Atomic Number (Z): Number of protons.
Mass Number (A): Protons + neutrons.
Neutrons: Mass number − atomic number.
Isotopes
Same element (same protons), different neutrons → different mass numbers.
Atomic symbol format: mass number (top left), atomic number (bottom left).
Atomic Mass
Weighted average of all naturally occurring isotopes; not a whole number.
Formula:
Bonus: Gases (Boyle's Law)
Boyle's Law
Boyle's Law describes the relationship between the pressure and volume of a gas at constant temperature. It states that the product of the initial pressure and volume is equal to the product of the final pressure and volume:
Formula:
To solve for final volume:
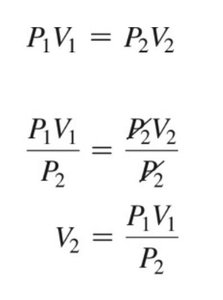
Example: If a gas at 2.0 atm occupies 3.0 L, what volume will it occupy at 1.0 atm? L.
