 Back
BackGibbs Energy and Thermodynamics: Spontaneity, Entropy, and Free Energy
Study Guide - Smart Notes

Gibbs Energy and Thermodynamics
Drivers of Physical and Chemical Change
Chemical and physical changes are driven by fundamental energetic and probabilistic factors. Thermodynamics helps us understand why certain processes occur spontaneously, while others do not.
Spontaneity in Chemical Systems
Spontaneity refers to the natural tendency of a process to occur without external intervention. Thermodynamics predicts whether a reaction is spontaneous, while kinetics describes how fast it occurs. A process can be spontaneous but extremely slow.
Spontaneous process: Occurs naturally under given conditions.
Nonspontaneous process: Requires continuous input of energy to proceed.
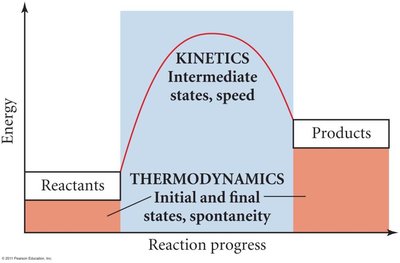
Thermodynamics vs. Kinetics
Thermodynamics determines the direction and extent of a reaction (spontaneity), while kinetics concerns the reaction rate (speed). Both are essential for understanding chemical change.
Spontaneity and Enthalpy
Many spontaneous reactions are exothermic (release heat, ΔH < 0), but not all. Some endothermic reactions (ΔH > 0) can also be spontaneous, indicating that enthalpy alone does not determine spontaneity.
Exothermic reactions: Often spontaneous, but not always.
Endothermic reactions: Can be spontaneous if other factors favor the process.
Entropy and the Second Law of Thermodynamics
Definition of Entropy (S)
Entropy is a measure of the disorder or randomness of a system. It quantifies the number of ways energy can be distributed among the particles of a system (microstates).
Higher entropy: More possible arrangements (microstates) of energy.
Lower entropy: Fewer possible arrangements.
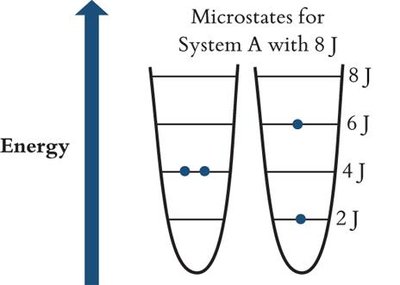
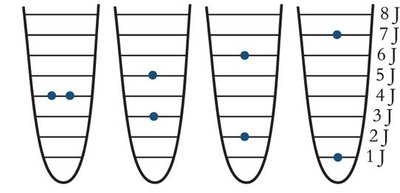
Energetically Equivalent States and Microstates
A macrostate (overall state) can be achieved through different microstates (specific arrangements of particles and energy). The macrostate with the highest number of microstates has the greatest entropy.
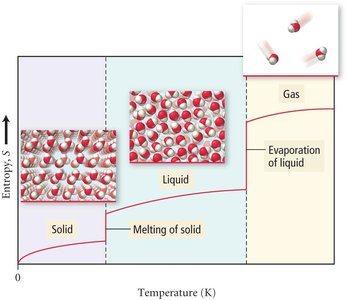
Entropy and Phase Changes
Entropy increases as matter changes from solid to liquid to gas, reflecting greater molecular motion and more ways to distribute energy.
Solid → Liquid → Gas: Entropy increases (ΔS > 0).
The Second Law of Thermodynamics
The second law states that for any spontaneous process, the entropy of the universe increases:
Spontaneous process: Increases the entropy of the universe.
Nonspontaneous process: Decreases or does not change the entropy of the universe.
Calculating Entropy Change
Entropy change for a process is a state function, depending only on the initial and final states:
Examples of Entropy Change
Water vapor condensing: Entropy decreases (ΔS < 0).
Sugar dissolving in tea: Entropy increases (ΔS > 0).
2NH3(g) → N2(g) + 3H2(g): Entropy increases (ΔS > 0).
Ag+(aq) + Cl−(aq) → AgCl(s): Entropy decreases (ΔS < 0).
Entropy Change in the Universe
For a process where the system's entropy decreases, the surroundings must increase in entropy by a greater amount for the process to be spontaneous.

Temperature Dependence of Entropy Change in Surroundings
The magnitude of entropy change in the surroundings is inversely proportional to temperature:
At higher temperatures, the same heat transfer causes a smaller change in entropy of the surroundings.
Standard Entropy and the Third Law of Thermodynamics
Standard Entropy Change (ΔS°rxn)
The standard entropy change for a reaction is the difference in entropy between products and reactants under standard conditions (1 bar, 298 K):
The Third Law of Thermodynamics
The entropy of a perfect crystal at absolute zero (0 K) is zero:
at for a perfect crystal.
Factors Affecting Entropy
Phase: Gases > Liquids > Solids
Molar mass: Higher molar mass, higher entropy (for similar types of substances).
Molecular complexity: More complex molecules have higher entropy.
Allotropes: Different forms of the same element (e.g., diamond vs. graphite) have different entropies.
Dissolution: Dissolving a solid increases entropy.
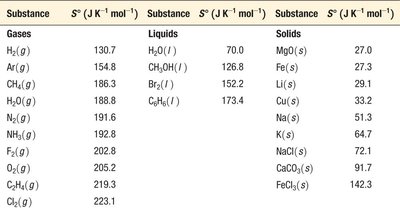

Relative Entropies: Table of Standard Molar Entropies
Substance | S° (J K−1 mol−1) |
|---|---|
H2(g) | 130.7 |
Ar(g) | 154.8 |
H2O(l) | 70.0 |
H2O(g) | 188.8 |
C(s, diamond) | 2.4 |
C(s, graphite) | 5.7 |
NO(g) | 210.8 |
NO2(g) | 240.1 |
N2O4(g) | 304.4 |
Gibbs Free Energy and Spontaneity
Definition of Gibbs Free Energy (G)
Gibbs free energy is the chemical potential energy available to do work at constant temperature and pressure. It combines enthalpy, entropy, and temperature into a single value:
Spontaneous process:
Nonspontaneous process:
Equilibrium:
Gibbs Free Energy Change for a Reaction
The change in Gibbs free energy for a reaction is given by:
If , the reaction is spontaneous.
If , the reaction is nonspontaneous.
Temperature Dependence of Spontaneity
The sign of can change with temperature, especially when and have the same sign. For example, the sublimation of dry ice (CO2(s)) is spontaneous at high temperature but not at low temperature.
ΔH | ΔS | Low Temperature | High Temperature | Example |
|---|---|---|---|---|
+ | + | Nonspontaneous (ΔG > 0) | Spontaneous (ΔG < 0) | 2H2O(s) → 2H2O(g) |
− | − | Spontaneous (ΔG < 0) | Nonspontaneous (ΔG > 0) | H2O(g) → H2O(l) |
+ | − | Nonspontaneous (ΔG > 0) | Nonspontaneous (ΔG > 0) | H2O(l) → H2O(s) |
− | + | Spontaneous (ΔG < 0) | Spontaneous (ΔG < 0) | 2H2(g) + O2(g) → 2H2O(g) |
Standard Gibbs Energy of Formation (ΔG°f)
The standard Gibbs energy of formation is the change in Gibbs energy when 1 mol of a compound forms from its elements in their standard states. For pure elements in their standard states, ΔG°f = 0.
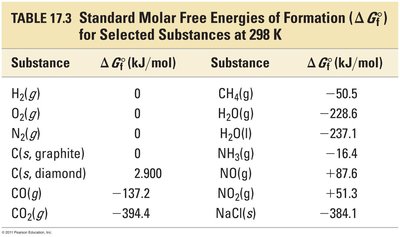
Hess's Law for Gibbs Energy
If a reaction can be expressed as the sum of a series of steps, the overall ΔG°rxn is the sum of the ΔG°rxn values for the individual steps.
Gibbs Energy at Non-Standard Conditions
The Gibbs energy change under non-standard conditions is given by:
Q is the reaction quotient, R is the gas constant, and T is temperature in Kelvin.
Gibbs Energy and Equilibrium
At equilibrium, ΔGrxn = 0. The relationship between standard Gibbs energy and the equilibrium constant K is:
Temperature Dependence of K: The van't Hoff Equation
The van't Hoff equation relates the equilibrium constant at two temperatures:
Summary Table: Key Equations
Equation | Description |
|---|---|
Total entropy change in the universe | |
Entropy change of surroundings | |
Gibbs free energy change | |
Gibbs energy at non-standard conditions | |
Relationship to equilibrium constant | |
van't Hoff equation |
Example: Calculate the standard entropy change for the reaction: 4NH3(g) + 5O2(g) → 4NO(g) + 6H2O(g) using standard molar entropies from the table above.
Additional info: These notes cover the core concepts of thermodynamics relevant to spontaneity, entropy, and Gibbs free energy, as outlined in a typical general chemistry curriculum.
