 Back
BackIdentifying the Correct Line Structure for a Branched Alkane
Study Guide - Smart Notes

Q3. Which of the following is a correct line structure for the given molecule?
Background
Topic: Organic Structure Representation (Alkanes)
This question tests your ability to interpret and match different representations of organic molecules, specifically branched alkanes. You need to recognize how condensed, line, and expanded structures correspond to each other.
Key Terms and Concepts:
Line Structure: A simplified way to draw organic molecules where carbon atoms are implied at the ends and bends of lines, and hydrogen atoms attached to carbons are not shown.
Branched Alkane: An alkane with one or more alkyl groups (branches) attached to the main carbon chain.
Condensed Structure: A way of writing organic molecules that shows all atoms but groups hydrogens with their attached carbons.
Step-by-Step Guidance
Carefully examine the condensed structure provided in the question. Identify the main carbon chain and locate all branches (alkyl groups) attached to it.
Count the total number of carbon atoms in the main chain and in each branch. Make sure to keep track of where each branch is attached.
For each line structure option, trace the main chain and check for the correct placement and number of branches. Remember that each vertex or end of a line represents a carbon atom.
Compare the connectivity (which carbon is bonded to which) in each line structure to the condensed structure. Eliminate any options that do not match the branching pattern or number of carbons.
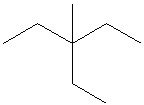
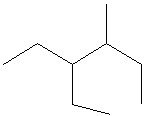
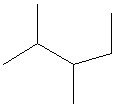
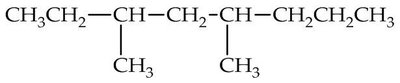
Try solving on your own before revealing the answer!
