 Back
BackIntermolecular Forces and Properties of Solids, Liquids, and Solutions
Study Guide - Smart Notes

Structure Determines Properties
Phases of Matter and Intermolecular Forces (IMFs)
The physical state of a substance—solid, liquid, or gas—is determined by the strength of intermolecular forces (IMFs) and the available thermal energy. IMFs are the attractions and repulsions between atoms and molecules, and their magnitude influences whether a substance is a gas, liquid, or solid at a given temperature.
Gas: Particles have complete freedom of motion, are far apart, and IMFs are very weak. Gases are compressible and expand to fill their container.
Liquid: Particles are closely packed but can move around, making liquids incompressible and able to flow and take the shape of their container. IMFs are stronger than in gases.
Solid: Particles are packed close together and fixed in position, making solids incompressible. IMFs are strongest in solids.
Example: The difference in molecular arrangement between liquid water and solid ice is due to the strength and type of IMFs present.
Intermolecular Forces (IMFs)
Types of IMFs
IMFs are not chemical bonds; they are much weaker than covalent or ionic bonds. However, they are crucial for determining melting points, boiling points, solubility, and the structure of biological molecules.
Ion-Dipole: Occurs between ions and polar molecules. Important for solubility of ionic compounds in water.
Dipole-Dipole: Occurs between polar molecules with permanent dipoles. Raises boiling and melting points compared to nonpolar molecules.
Hydrogen Bonding: A particularly strong dipole-dipole interaction involving H bonded to N, O, or F. Responsible for unique properties of water and biological molecules like DNA.
London Dispersion (Van der Waals): Present in all molecules, especially nonpolar ones. Strength increases with molar mass and polarizability.
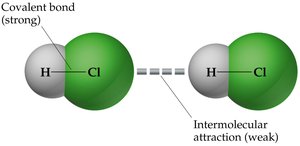
Example: The energy required to break a covalent bond (e.g., H–Cl) is much greater than the energy required to overcome IMFs between molecules.
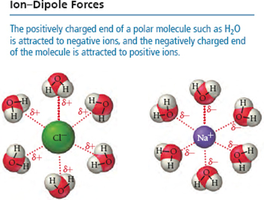
Ion-Dipole Interactions
Ion-dipole forces are the strongest type of IMF and occur when ions interact with polar molecules. The strength of these interactions is a major factor in the solubility of ionic compounds in water.
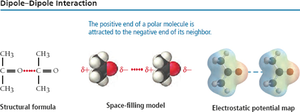
Dipole-Dipole Interactions
Dipole-dipole forces occur between polar molecules. The positive end of one molecule is attracted to the negative end of another, increasing boiling and melting points.
Hydrogen Bonding
Hydrogen bonding is a strong type of dipole-dipole interaction. It occurs when hydrogen is bonded to highly electronegative atoms (N, O, F). This leads to higher boiling and melting points and is essential for the structure of water, DNA, and proteins.
Example: Water's high boiling point and ice's lower density compared to liquid water are due to hydrogen bonding.
London Dispersion Forces
London dispersion forces arise from temporary fluctuations in electron distribution, creating instantaneous dipoles. These forces are present in all molecules and increase with molar mass and polarizability.
Example: Iodine (I2) is more polarizable than chlorine (Cl2), resulting in stronger dispersion forces.
Properties Associated with the Liquid State
Viscosity, Surface Tension, and Capillary Action
IMFs influence several properties of liquids:
Viscosity: Resistance to flow. Stronger IMFs and higher molar mass increase viscosity; higher temperature decreases it.
Surface Tension: Tendency of liquids to minimize surface area. Stronger IMFs result in higher surface tension.
Capillary Action: Movement of liquid up a thin tube due to adhesive and cohesive forces.
Phase Transitions and Energetics
Vaporization, Boiling Point, and Vapor Pressure
Vaporization occurs when molecules at the surface of a liquid gain enough energy to escape into the gas phase. The boiling point is the temperature at which vapor pressure equals external pressure. Stronger IMFs result in lower vapor pressure and higher boiling points.
Heating Curves and Phase Diagrams
Heating curves show temperature changes during phase transitions. Phase diagrams map the states of matter at various temperatures and pressures, including critical and triple points.
Solids and Crystal Structures
Crystalline vs. Amorphous Solids
Solids are classified as crystalline (ordered, repeating patterns) or amorphous (disordered, no long-range order). Crystalline solids are studied using X-ray crystallography.
Unit Cells and Lattice Structures
The unit cell is the smallest repeating unit in a crystal lattice. Cubic unit cells include primitive (simple cubic), body-centered cubic (BCC), and face-centered cubic (FCC), each with a distinct arrangement and number of atoms per cell.
Types of Solids and Modern Materials
Classification of Solids
Molecular Solids: Held together by IMFs; low melting points.
Ionic Solids: Held together by ionic bonds; high melting points.
Metallic Solids: Held together by metallic bonds; variable melting points.
Network Covalent Solids: Held together by covalent bonds; very high melting points.
Nonbonding Atomic Solids: Held together by dispersion forces; very low melting points.
Solutions and Solubility
Types of Solutions and Solubility
Solutions are homogeneous mixtures of solute and solvent. Solubility depends on IMFs: "like dissolves like"—polar solutes dissolve in polar solvents, nonpolar in nonpolar.
Energetics of Solution Formation
Formation of solutions involves breaking solute-solute and solvent-solvent interactions and forming solute-solvent interactions. The enthalpy change () determines whether the process is endothermic or exothermic.
Colligative Properties
Colligative properties depend on the number of solute particles, not their identity. These include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.
Boiling Point Elevation:
Freezing Point Depression:
Osmotic Pressure:
van't Hoff Factor
The van't Hoff factor () accounts for the number of particles produced by ionic solutes. For example, NaCl produces 2 particles per formula unit.
Summary Table: Types of Intermolecular Forces
Type of IMF | Particles Involved | Relative Strength | Example |
|---|---|---|---|
Ion-Dipole | Ion & Polar Molecule | Strongest | Na+ in water |
Hydrogen Bonding | H bonded to N, O, F | Very Strong | Water, DNA |
Dipole-Dipole | Polar Molecules | Moderate | HCl, formaldehyde |
London Dispersion | All molecules | Weakest | I2, Cl2 |
Key Equations
Clausius-Clapeyron Equation:
Henry's Law:
Osmotic Pressure:
Conclusion
Understanding intermolecular forces is essential for predicting the properties of solids, liquids, and solutions. These forces determine phase behavior, solubility, and many physical properties critical to chemistry and biology.
