 Back
BackIntermolecular Forces and States of Matter: Study Guide
Study Guide - Smart Notes

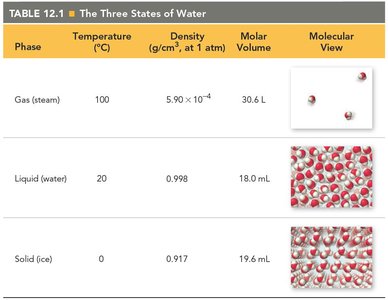
The Three States of Water
Physical Properties and Molecular Structure
Water exists in three primary states—gas, liquid, and solid—each with distinct physical properties and molecular arrangements. Understanding these states is fundamental to general chemistry, as it illustrates the effects of intermolecular forces on matter.
Gas (Steam): Molecules are far apart, low density, and have indefinite shape and volume.
Liquid (Water): Molecules are closer together, higher density, indefinite shape but definite volume.
Solid (Ice): Molecules are tightly packed, high density, definite shape and volume.
Phase | Temperature (°C) | Density (g/cm3, at 1 atm) | Molar Volume | Molecular View |
|---|---|---|---|---|
Gas (steam) | 100 | 5.90 × 10−4 | 30.6 L | Molecules far apart |
Liquid (water) | 20 | 0.998 | 18.0 mL | Molecules closer together |
Solid (ice) | 0 | 0.917 | 19.6 mL | Molecules tightly packed |
Properties of the States of Matter
The physical properties of matter—density, shape, volume, and strength of intermolecular forces—vary between gas, liquid, and solid states. These properties are determined by the arrangement and interactions of molecules.
State | Density | Shape | Volume | Strength of Intermolecular Forces |
|---|---|---|---|---|
Gas | Low | Indefinite | Indefinite | Weak |
Liquid | High | Indefinite | Definite | Moderate |
Solid | High | Definite | Definite | Strong |
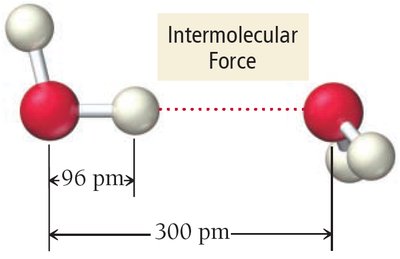
Intermolecular Forces
Definition and Importance
Intermolecular forces are the forces of attraction or repulsion between neighboring molecules. They originate from interactions among charges, partial charges, and temporary charges. These forces are responsible for many physical properties, such as melting point, boiling point, and vapor pressure.
Coulomb’s Law: The potential energy (E) between two oppositely charged particles is given by: where and are the charges, is the separation, and is the permittivity of free space.
Stronger intermolecular forces require more energy to separate molecules.
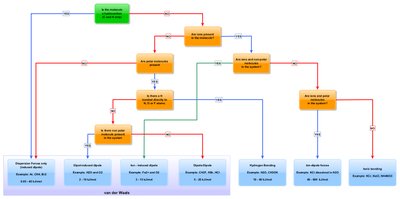
Types of Intermolecular Forces
There are several types of intermolecular forces, each with unique characteristics and effects on molecular behavior.
London Dispersion Forces (LDF): Temporary attractions due to momentary uneven electron distribution.
Dipole-Dipole Forces: Attractions between polar molecules.
Hydrogen Bonding: Special strong dipole-dipole interaction involving H bonded to F, O, or N.
Ion-Dipole Forces: Attractions between ions and polar molecules.
Induced Dipole Forces: Attractions caused by the proximity of a polar molecule or ion to a nonpolar molecule.
London Dispersion Forces (LDF)
Origin and Characteristics
London Dispersion Forces arise from temporary dipoles created by fluctuations in electron distribution. These forces are present in all molecules and atoms, but are especially significant in nonpolar molecules.
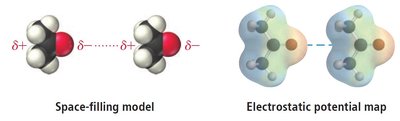
Temporary dipoles induce dipoles in neighboring molecules.
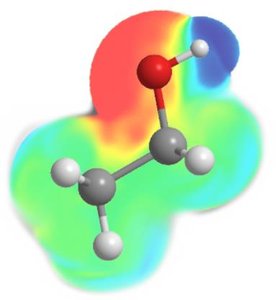
Regions with excess electron density have partial negative charge (), while regions with depleted electron density have partial positive charge ().
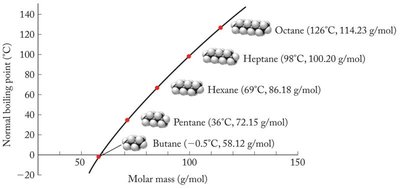
Larger atoms or molecules are more polarizable, leading to stronger dispersion forces.


Polarizability and Molecular Shape
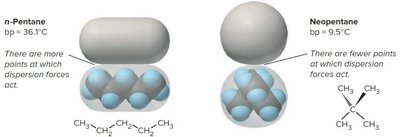
The strength of dispersion forces increases with polarizability and surface area. Linear molecules generally have higher dispersion forces than branched or bulky molecules of similar molecular weight.
Polarizability: The ease with which the electron cloud of a molecule can be distorted.
Surface Area: Greater surface area increases intermolecular interactions.
Example: Pentane (linear) has a higher boiling point than 2,2-dimethylpropane (branched) due to greater dispersion forces.


Dipole-Dipole Forces
Definition and Effects
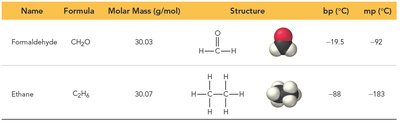
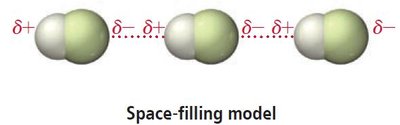
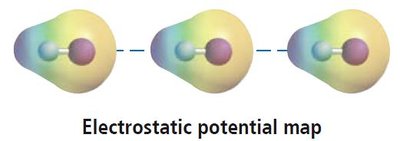
Dipole-dipole forces occur between polar molecules, where the positive end of one molecule is attracted to the negative end of another. These forces are stronger than dispersion forces and significantly affect boiling and melting points.
All molecules have dispersion forces, but only polar molecules have dipole-dipole forces.
Larger dipole moments lead to stronger dipole-dipole interactions.
Example: Formaldehyde (CH2O) has a higher boiling point than ethane (C2H6) due to dipole-dipole forces.

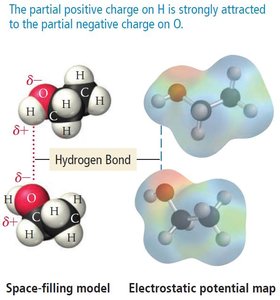
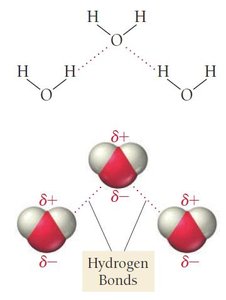
Hydrogen Bonding
Definition and Importance
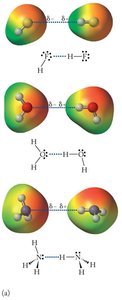
Hydrogen bonding is a special type of dipole-dipole interaction that occurs when hydrogen is bonded directly to a highly electronegative atom (F, O, or N). Hydrogen bonds are much stronger than typical dipole-dipole forces, but weaker than covalent bonds.
Hydrogen bonds are responsible for many unique properties of water, such as high boiling point and surface tension.
A typical hydrogen bond is only 2–5% as strong as a covalent bond.
Examples and Applications
Hydrogen bonding is crucial in biological systems (e.g., DNA base pairing) and in determining the physical properties of substances.
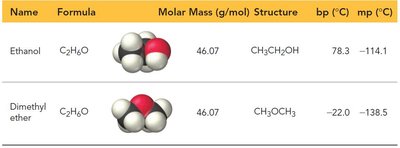
Example: Ethanol (C2H5OH) has a much higher boiling point than dimethyl ether (CH3OCH3) due to hydrogen bonding.
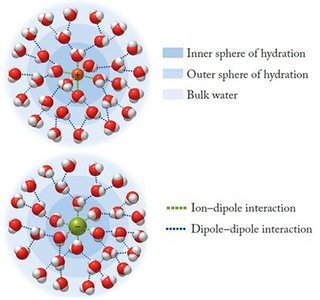
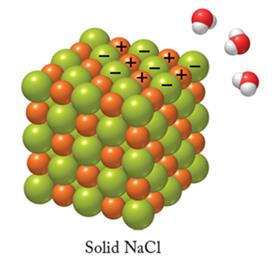
Ion–Dipole Forces
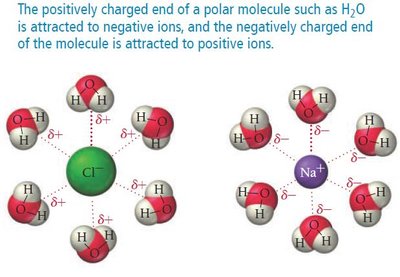
Definition and Role in Solubility
Ion–dipole forces occur in mixtures where ions from an ionic compound are attracted to the dipole of polar molecules. These forces are a key factor in the solubility of ionic compounds in water.
Water molecules surround ions, forming a sphere of hydration.
If the solvent is not water, the cluster is called a sphere of solvation.



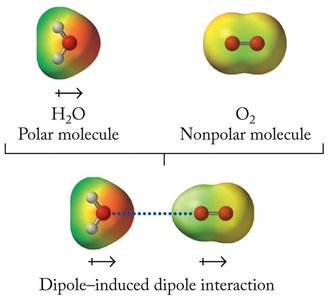
Induced Dipole Forces
Definition and Mechanism
Induced dipole forces arise when the proximity of a polar molecule or ion causes a temporary dipole in a nonpolar molecule. This interaction is weaker than other intermolecular forces but still significant in certain contexts.
Polar molecules can induce dipoles in nonpolar molecules, leading to attraction.
Example: Water (polar) can induce a dipole in oxygen (nonpolar).


Additional info: Academic context was added to clarify the definitions, mechanisms, and examples of each type of intermolecular force, as well as to explain the physical properties of the states of matter and their dependence on molecular interactions.
