 Back
BackIntermolecular Forces and Their Effects on Physical Properties
Study Guide - Smart Notes

Intermolecular Forces
Introduction to Intermolecular Forces
Intermolecular forces are the attractive forces that exist between molecules, influencing the physical properties of substances such as boiling point, melting point, viscosity, and surface tension. These forces are generally weaker than intramolecular (covalent or ionic) bonds but are crucial in determining the behavior of liquids and solids.
Hydrogen Bonding
Definition and Nature of Hydrogen Bonds
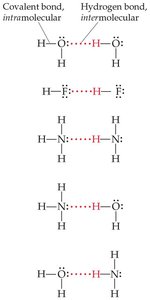
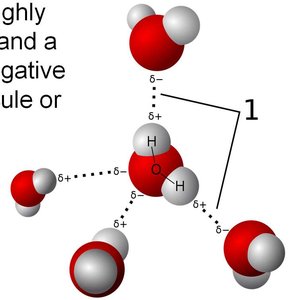
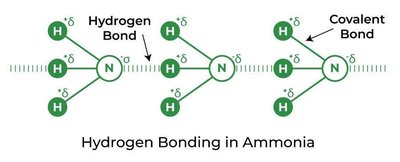
Hydrogen bonding is a special type of dipole–dipole interaction that occurs when hydrogen is covalently bonded to highly electronegative atoms such as nitrogen (N), oxygen (O), or fluorine (F). The hydrogen atom, having no inner electrons, interacts strongly with lone pairs on nearby electronegative atoms in other molecules, resulting in an unusually strong attraction.
Hydrogen bond: Attraction between a hydrogen atom attached to N, O, or F and a lone pair on another N, O, or F atom.
Hydrogen bonds are much stronger than typical dipole–dipole interactions but weaker than covalent bonds.
What Forms Hydrogen Bonds?
Hydrogen bonding arises due to the high electronegativity of N, O, and F, which creates a significant partial positive charge on the hydrogen atom. This nearly bare proton is strongly attracted to lone pairs on electronegative atoms in neighboring molecules.
Hydrogen bonds are only possible when H is directly bonded to N, O, or F.
There must be a nonbonding pair of electrons on the electronegative atom for hydrogen bonding to occur.
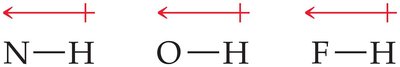
Examples and Applications of Hydrogen Bonding
Water (H2O): Extensive hydrogen bonding leads to high boiling and melting points, and unique properties such as ice being less dense than liquid water.
Ammonia (NH3): Hydrogen bonding occurs between N-H groups.
DNA: Hydrogen bonds between base pairs stabilize the double helix structure, ensuring accurate genetic information transfer.

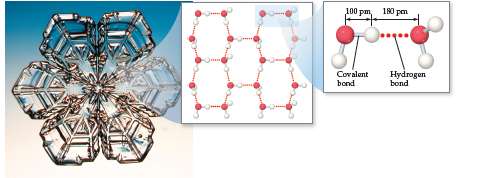
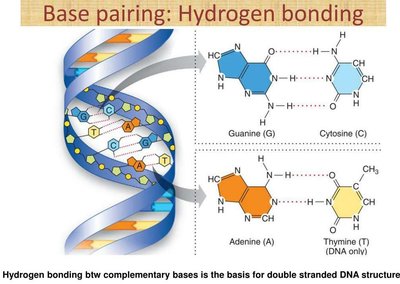
Identifying Substances Capable of Hydrogen Bonding
To determine if a substance can form hydrogen bonds, check for H bonded to N, O, or F, and the presence of lone pairs on these atoms in neighboring molecules. For example, hydrazine (H2NNH2) can form hydrogen bonds, while methane (CH4), methyl fluoride (CH3F), and hydrogen sulfide (H2S) cannot.
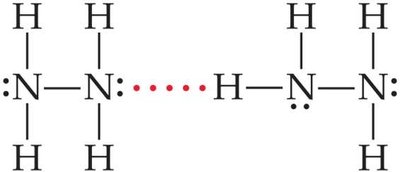
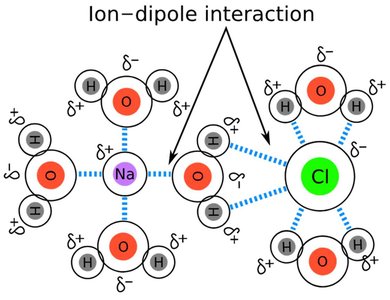
Ion–Dipole Interactions
Definition and Importance
Ion–dipole interactions occur between an ion and a polar molecule. These forces are especially important in solutions of ionic compounds in polar solvents, such as salts dissolving in water. The strength of ion–dipole interactions helps overcome the lattice energy of ionic solids, allowing them to dissolve.
Essential for the dissolution of ionic substances in water.
Strength depends on the charge and size of the ion and the dipole moment of the solvent molecule.

Types and Strengths of Intermolecular Forces
Classification of Intermolecular Forces
There are several types of intermolecular forces, each with different strengths and characteristics:
Type of Intermolecular Interaction | Atoms | Nonpolar Molecules | Polar Molecules without OH, NH, or HF | Polar Molecules with OH, NH, or HF | Ionic Solids in Polar Liquids |
|---|---|---|---|---|---|
Dispersion Forces (0.1–30 kJ/mol) | ✓ | ✓ | ✓ | ✓ | |
Dipole–Dipole Interactions (2–15 kJ/mol) | ✓ | ✓ | |||
Hydrogen Bonding (10–40 kJ/mol) | ✓ | ||||
Ion–Dipole Interactions (>50 kJ/mol) | ✓ |
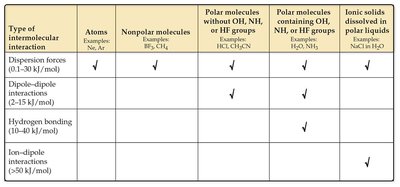
Generalizations about Relative Strengths
Dispersion forces are present in all substances.
The strongest intermolecular force present determines the substance's physical properties.
For molecules with similar molar masses and shapes, dispersion forces are comparable.
For molecules with very different molar masses and no hydrogen bonding, dispersion forces dominate.
Physical Properties Affected by Intermolecular Forces
Boiling and Melting Points
The boiling and melting points of substances are directly related to the strength of intermolecular forces. Stronger forces result in higher boiling and melting points.
Ionic substances have the highest boiling points due to strong ionic bonds.
Hydrogen bonding increases boiling points compared to substances with only dipole–dipole or dispersion forces.
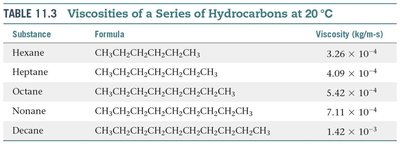
Viscosity
Viscosity is the resistance of a liquid to flow. It increases with stronger intermolecular forces and decreases with higher temperature. Longer molecular chains also increase viscosity due to greater surface area and entanglement.
Measured by timing flow through a tube or the rate at which objects fall through the liquid.
Viscosity decreases as temperature increases because higher kinetic energy overcomes attractive forces.

Substance | Formula | Viscosity (kg/m·s) |
|---|---|---|
Hexane | CH3CH2CH2CH2CH2CH3 | 3.26 × 10–4 |
Heptane | CH3CH2CH2CH2CH2CH2CH3 | 4.09 × 10–4 |
Octane | CH3CH2CH2CH2CH2CH2CH2CH3 | 5.42 × 10–4 |
Nonane | CH3CH2CH2CH2CH2CH2CH2CH2CH3 | 7.11 × 10–4 |
Decane | CH3CH2CH2CH2CH2CH2CH2CH2CH2CH3 | 1.42 × 10–3 |
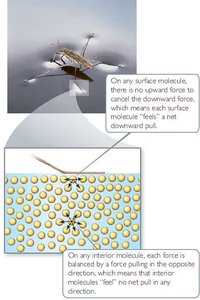
Surface Tension
Surface tension is the energy required to increase the surface area of a liquid. It results from extra inward forces on surface molecules, causing liquids like water to bead up on nonpolar surfaces.
Cohesion and Adhesion
Cohesive forces bind similar molecules together, while adhesive forces bind molecules to different substances or surfaces. These forces are important in phenomena such as capillary action.
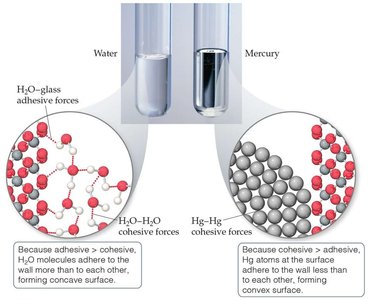
Capillary Action
Capillary action is the rise of liquids in narrow tubes due to adhesive and cohesive forces. Water rises in glass due to strong adhesive forces, while mercury does not due to stronger cohesive forces within itself.
Phase Changes
Types of Phase Changes
A phase change is the conversion from one state of matter to another, involving energy transfer. Common phase changes include melting (fusion), freezing, vaporization, condensation, sublimation, and deposition.
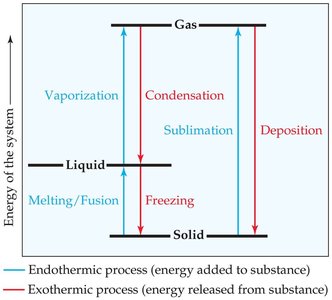
Energy Changes in Phase Transitions
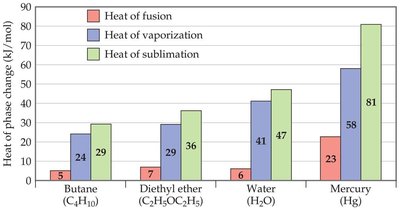
Heat of fusion (ΔHfus): Energy required to change a solid to a liquid at its melting point.
Heat of vaporization (ΔHvap): Energy required to change a liquid to a gas at its boiling point.
Heat of sublimation (ΔHsub): Energy required to change a solid directly to a gas.
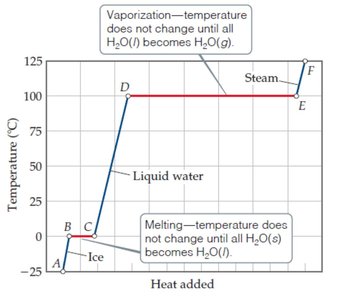
Heating Curves
A heating curve is a graph of temperature versus heat added, showing the temperature changes and phase transitions as a substance is heated. During phase changes, temperature remains constant while energy is used to change the phase.
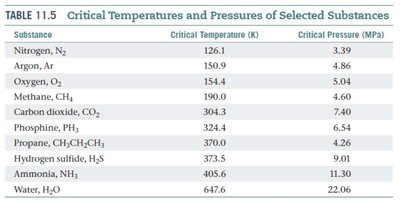
Critical Temperature and Supercritical Fluids
The critical temperature is the highest temperature at which a substance can exist as a liquid, regardless of pressure. Above this temperature and the corresponding critical pressure, the liquid and gas phases become indistinguishable, forming a supercritical fluid with unique solvent properties.
Substance | Critical Temperature (K) | Critical Pressure (MPa) |
|---|---|---|
Nitrogen, N2 | 126.1 | 3.39 |
Argon, Ar | 150.9 | 4.86 |
Oxygen, O2 | 154.4 | 5.04 |
Methane, CH4 | 190.0 | 4.60 |
Water, H2O | 647.6 | 22.06 |
Ammonia, NH3 | 405.6 | 11.30 |
Vapour Pressure
Definition and Temperature Dependence
Vapour pressure is the pressure exerted by a vapor in equilibrium with its liquid at a given temperature. As temperature increases, more molecules have enough kinetic energy to escape the liquid phase, increasing vapor pressure.
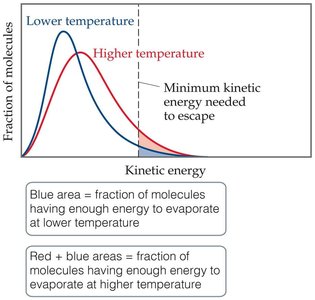
Dynamic Equilibrium and Boiling Point
When the rate of evaporation equals the rate of condensation, the system is at dynamic equilibrium. The boiling point is the temperature at which the vapor pressure equals atmospheric pressure. The normal boiling point is defined at 760 torr (101.3 kPa).
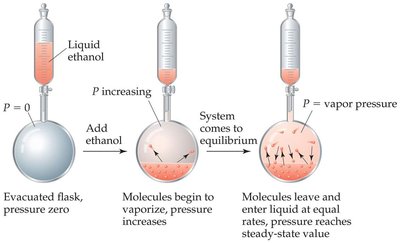

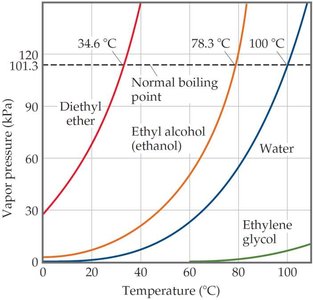
Relating Boiling Point to Vapor Pressure
To determine the boiling point at a given pressure, locate the pressure on a vapor pressure curve and find the corresponding temperature. For example, diethyl ether boils at about 27°C under 0.80 atm (81 kPa) pressure.
Additional info: The concepts of intermolecular forces and their effects on physical properties are foundational for understanding the behavior of liquids and solids, as well as many biological and industrial processes.
