 Back
BackIntermolecular Forces and Their Effects on Properties of Solids, Liquids, and Solutions
Study Guide - Smart Notes

Structure Determines Properties
Phases of Matter and Intermolecular Forces
The physical state of a substance (solid, liquid, or gas) is determined by the magnitude of its intermolecular forces (IMFs) and the available thermal energy. IMFs are the attractions and repulsions between atoms and molecules, and their strength dictates whether a substance exists as a solid, liquid, or gas under given conditions.
Gas: Particles have complete freedom of motion, are far apart, and IMFs are very weak. Gases are compressible and expand to fill their container.
Liquid: Particles are closely packed but can move around. IMFs are stronger than in gases, making liquids incompressible and able to flow.
Solid: Particles are packed closely together in fixed positions. IMFs are strongest, making solids incompressible and often crystalline.
Example: Water exists as ice (solid), liquid water, or steam (gas) depending on temperature and IMFs.
Intermolecular Forces (IMFs)
IMFs vs. Chemical Bonds
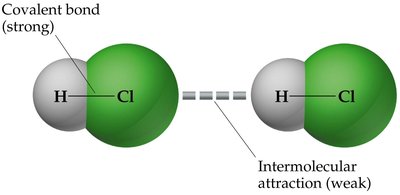
IMFs are much weaker than the covalent or ionic bonds that hold atoms together within molecules. However, they are crucial for determining melting points, boiling points, solubility, and the structure of biological molecules.
Covalent bonds: Strong, hold atoms together within molecules.
IMFs: Weak, hold molecules together in condensed phases.

Types of Intermolecular Forces
The type and strength of IMFs depend on three properties:
Charge (ion or not)
Polarity (molecular shape, dipoles)
Molar mass (size, polarizability)
Major types of IMFs:
Ion-Dipole: Attraction between ions and polar molecules.
Dipole-Dipole: Attraction between polar molecules.
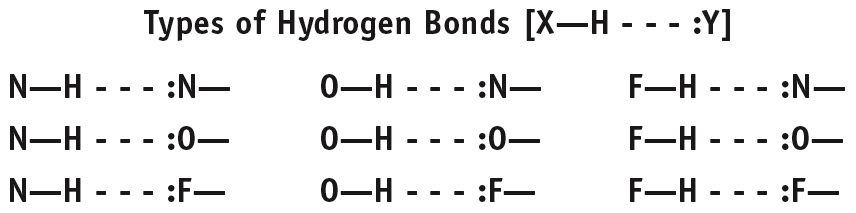
Hydrogen Bonding: Strong dipole-dipole interaction involving H bonded to N, O, or F.
London Dispersion: Attraction due to temporary dipoles in all molecules and atoms.
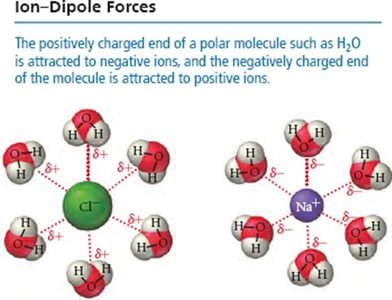
Ion-Dipole Interactions
Ion-dipole forces occur when ions from an ionic compound are attracted to the dipole of polar molecules. This is a key factor in the solubility of ionic compounds in water.
Dipole-Dipole Forces
Polar molecules have permanent dipoles and interact via dipole-dipole forces. The strength of these forces increases with the polarity of the molecules, raising boiling and melting points compared to nonpolar molecules of similar size.
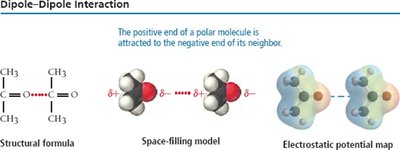
Example: Formaldehyde vs. Ethane
Formaldehyde (polar, dipole-dipole): Higher boiling and melting points.
Ethane (nonpolar, dispersion only): Lower boiling and melting points.

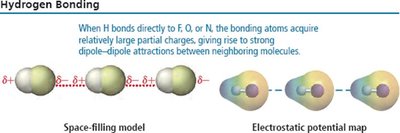
Hydrogen Bonding
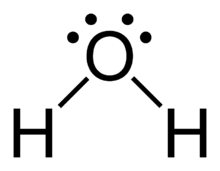
Hydrogen bonding is a particularly strong type of dipole-dipole interaction, occurring when H is bonded to N, O, or F. It significantly affects the properties of water, DNA, and proteins.
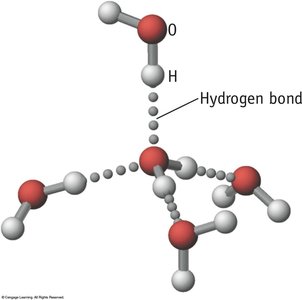
Hydrogen Bonding in Water
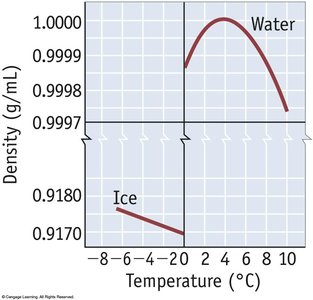
Hydrogen bonds create a network of interactions in water, leading to phenomena such as capillary action, surface tension, and the flotation of ice. Ice has an open lattice structure, making it less dense than liquid water.
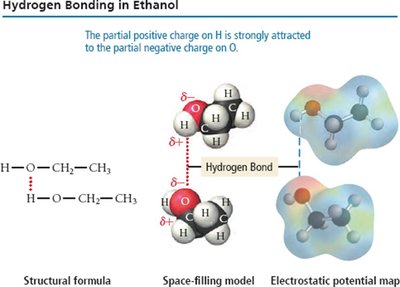
Hydrogen Bonding in Ethanol
Ethanol molecules form hydrogen bonds, resulting in higher boiling and melting points compared to similar molecules without hydrogen bonding.
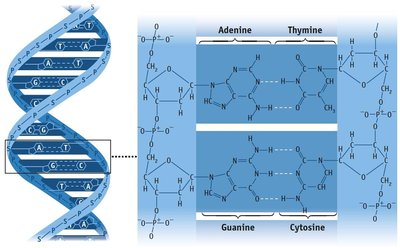
Hydrogen Bonding in DNA
Hydrogen bonds are responsible for the base pairing in DNA, holding the double helix together.
London Dispersion Forces
Dispersion forces arise from temporary fluctuations in electron distribution, creating instantaneous dipoles. These forces are present in all molecules and atoms, but are the only IMFs in nonpolar molecules.

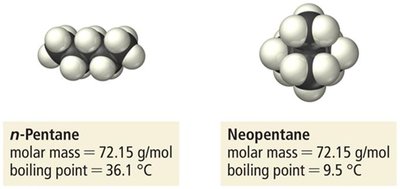
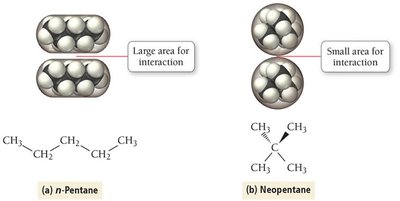
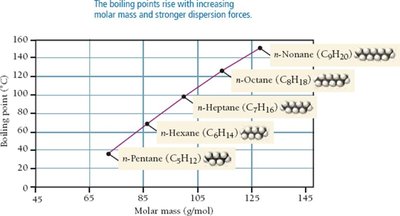
Effect of Molar Mass and Shape
As molar mass increases, dispersion forces become stronger due to greater polarizability. Molecular shape also affects dispersion force strength; straight-chain molecules have higher boiling points than branched isomers due to greater surface contact.
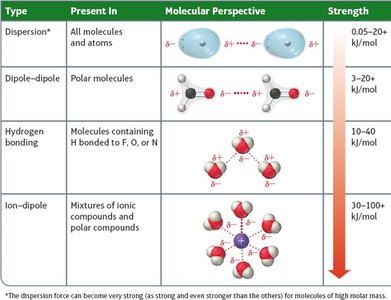
Summary Table: Types of Intermolecular Forces
Type | Present In | Molecular Perspective | Strength |
|---|---|---|---|
Dispersion | All molecules and atoms | Temporary dipoles | 0.05–20 kJ/mol |
Dipole-Dipole | Polar molecules | Permanent dipoles | 3–20 kJ/mol |
Hydrogen Bonding | Molecules with H bonded to F, O, or N | Strong dipole-dipole | 10–40 kJ/mol |
Ion-Dipole | Mixtures of ions and polar compounds | Ion and dipole interaction | 30–100+ kJ/mol |
IMFs and Solubility
Solubility and Miscibility
Solubility depends on the attractive forces between solute and solvent molecules. "Like dissolves like": polar substances dissolve in polar solvents, nonpolar substances dissolve in nonpolar solvents. Miscible liquids mix without separating; immiscible liquids do not mix.
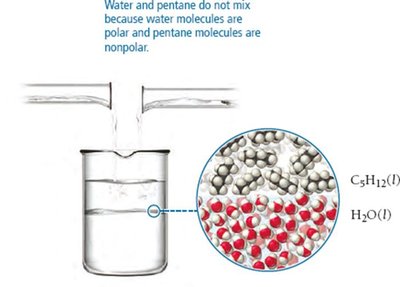
Induced Dipole Forces
Polar molecules can induce a dipole in nonpolar molecules, allowing them to mix. For example, water (polar) can induce a dipole in O2 (nonpolar), enabling oxygen to dissolve in water.
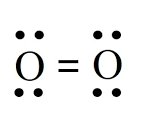
Properties of Liquids
Viscosity
Viscosity is the resistance of a liquid to flow. It increases with stronger IMFs, higher molar mass, and decreases with increasing temperature.
Surface Tension
Surface tension is the tendency of liquids to minimize their surface area, forming a "skin" due to tightly packed surface molecules. Stronger IMFs lead to higher surface tension.
Capillary Action
Capillary action is the movement of a liquid up a thin tube, resulting from adhesive forces (attraction to the tube) and cohesive forces (attraction between liquid molecules).
Phase Transitions
Vaporization and Vapor Pressure
Vaporization is the process by which molecules escape from the liquid phase to the gas phase. The vapor pressure is the pressure exerted by the vapor when equilibrium is established between vaporization and condensation.
Boiling Point
The boiling point is the temperature at which the vapor pressure equals the external pressure. Stronger IMFs result in higher boiling points.
Melting (Fusion) and Heat of Fusion
Melting is the transition from solid to liquid, requiring energy to overcome IMFs. The heat of fusion is the energy required to melt one mole of solid.
Phase Diagrams
Phase diagrams show the states of matter at various temperatures and pressures, including regions for solid, liquid, and gas, and lines for phase transitions.
Crystalline Solids and Unit Cells
Types of Solids
Crystalline: Well-ordered, definite arrangement; melts at specific temperatures.
Amorphous: No orderly arrangement; no specific melting point.
Unit Cells
The unit cell is the smallest repeating unit in a crystal lattice. Cubic unit cells include primitive (simple cubic), body-centered cubic (BCC), and face-centered cubic (FCC).
Primitive: 1 atom per unit cell.
BCC: 2 atoms per unit cell.
FCC: 4 atoms per unit cell.
Types of Crystalline Solids
Classification
Molecular solids: Molecules held by IMFs; low melting points.
Ionic solids: Ions held by electrostatic attraction; high melting points.
Metallic solids: Atoms held by metallic bonds; variable melting points.
Network covalent solids: Atoms held by covalent bonds; very high melting points.
Nonbonding atomic solids: Noble gases held by dispersion forces; very low melting points.
Solutions and Solution Properties
Types of Solutions
Solutions are homogeneous mixtures of solute and solvent. Types include gaseous, liquid, and solid solutions.
Solubility
Solubility is the maximum amount of solute that can dissolve in a solvent. Miscible liquids mix completely; immiscible liquids do not.
Concentration Units
Molarity (M): Moles of solute per liter of solution.
Molality (m): Moles of solute per kilogram of solvent.
Weight percent: Mass of solute per mass of solution, times 100%.
Mole fraction (X): Moles of solute divided by total moles.
Colligative Properties
Vapor Pressure Lowering
The vapor pressure of a solvent decreases when a nonvolatile solute is added. Raoult’s Law describes this:
Boiling Point Elevation
Adding solute raises the boiling point:
Freezing Point Depression
Adding solute lowers the freezing point:
Osmotic Pressure
Osmosis is the flow of solvent through a semipermeable membrane. The osmotic pressure is given by:
van’t Hoff Factor
For ionic solutes, the van’t Hoff factor (i) accounts for the number of particles produced per formula unit:
Colloids and Surfactants
Colloids
Colloids are mixtures with dispersed particles larger than those in true solutions but smaller than those in suspensions. They do not settle out.
Surfactants
Surfactants have both hydrophilic and hydrophobic regions, allowing them to interact with both water and nonpolar substances. Soaps and detergents are common surfactants.
