 Back
BackIntermolecular Forces and Their Role in the Properties of Solids, Liquids, and Solutions
Study Guide - Smart Notes

Structure Determines Properties
Phases of Matter and Molecular Arrangement
The physical state of a substance—solid, liquid, or gas—is determined by the strength and type of intermolecular forces (IMFs) present, as well as the available thermal energy. IMFs are responsible for the existence of condensed states (solids and liquids) and influence properties such as density, compressibility, and molecular arrangement.
Gas: Molecules are far apart, move freely, and IMFs are very weak. Gases are compressible and fill their container.
Liquid: Molecules are close together but can move past each other. IMFs are stronger than in gases, making liquids incompressible and able to flow.
Solid: Molecules are tightly packed in fixed positions. IMFs are strongest, making solids incompressible and rigid.
Intermolecular Forces (IMFs)
IMFs vs. Chemical Bonds
IMFs are much weaker than covalent or ionic bonds, but they play a crucial role in determining melting points, boiling points, solubility, and the structure of biological molecules.
Covalent bonds: Strong, hold atoms together within molecules.
IMFs: Weak, act between molecules or ions.
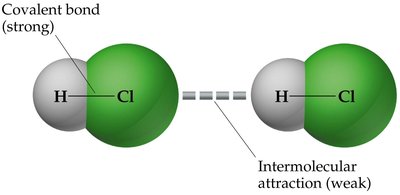

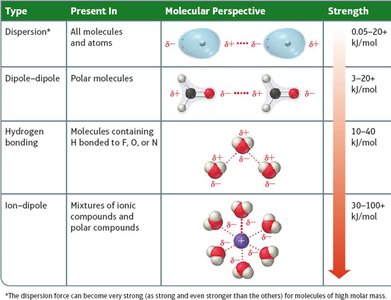
Types of Intermolecular Forces
The type and strength of IMFs depend on three main properties:
Presence of charge (ion-dipole)
Molecular polarity (dipole-dipole, hydrogen bonding)
Molar mass and polarizability (London dispersion)
Ion-Dipole Forces
Occur between ions and polar molecules. These are especially important in the solubility of ionic compounds in water.
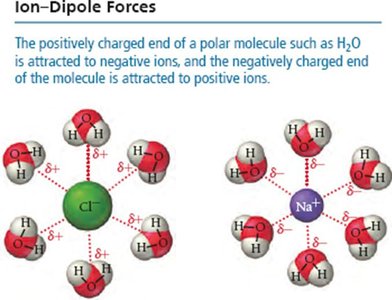
Dipole-Dipole Forces
Present in polar molecules, where the positive end of one molecule is attracted to the negative end of another. These forces increase boiling and melting points compared to nonpolar molecules of similar size.
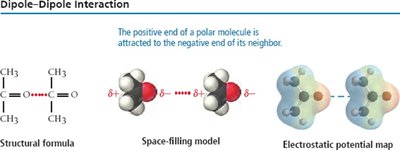

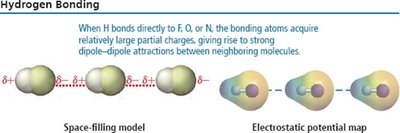
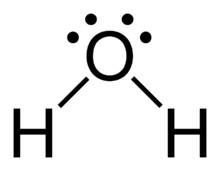
Hydrogen Bonding
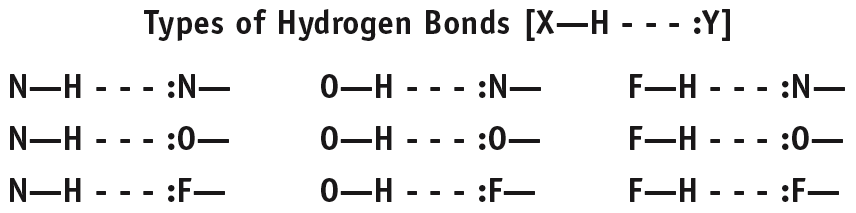
A particularly strong type of dipole-dipole interaction, occurring when hydrogen is bonded to N, O, or F. Hydrogen bonds significantly affect physical properties such as boiling point, melting point, and solubility.
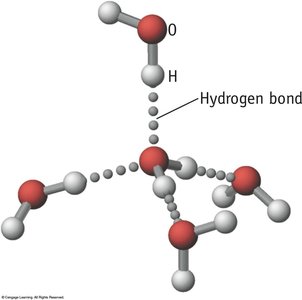
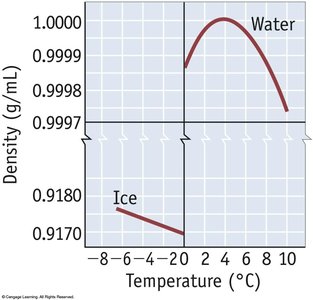
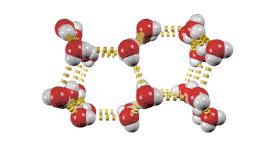
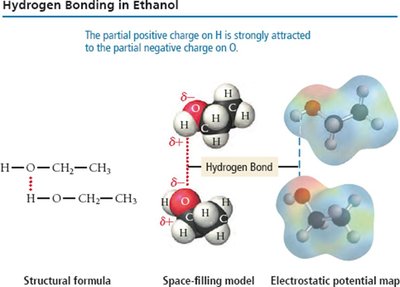
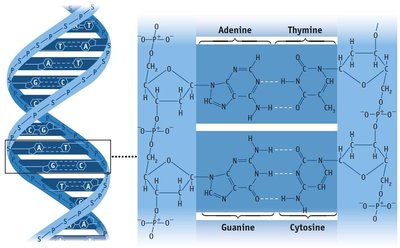
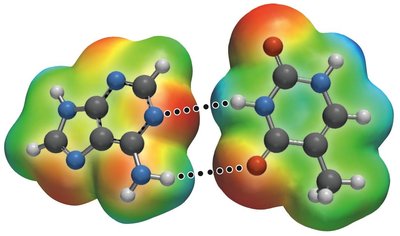
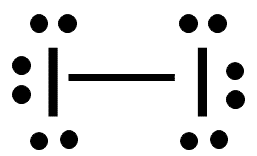
London Dispersion Forces
Present in all molecules and atoms, but are the only IMFs in nonpolar substances. These forces arise from temporary fluctuations in electron distribution, creating instantaneous dipoles.

Effect of Molar Mass and Shape on Dispersion Forces
As molar mass increases, dispersion forces become stronger due to greater polarizability. Molecular shape also affects the magnitude of dispersion forces; straight-chain molecules have higher boiling points than branched ones due to greater surface contact.
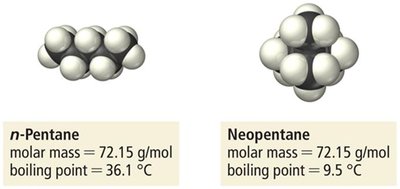
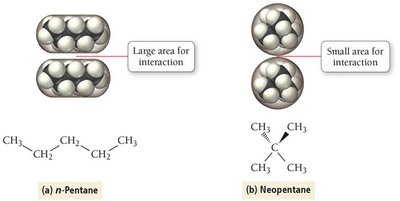
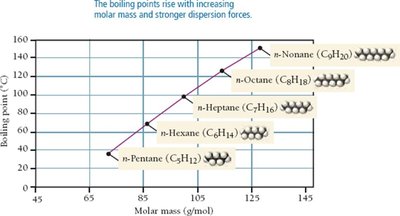
Summary Table of Intermolecular Forces
Type | Present In | Molecular Perspective | Strength |
|---|---|---|---|
Dispersion | All molecules and atoms | Instantaneous dipoles | 0.05–20 kJ/mol |
Dipole–Dipole | Polar molecules | Permanent dipoles | 3–20 kJ/mol |
Hydrogen Bonding | Molecules with H bonded to F, O, or N | Strong dipole–dipole | 10–40 kJ/mol |
Ion–Dipole | Mixtures of ions and polar compounds | Ion and dipole interaction | 30–100+ kJ/mol |
IMFs and Solubility
Solubility and Miscibility
Solubility depends on the compatibility of IMFs between solute and solvent. "Like dissolves like"—polar substances dissolve in polar solvents, nonpolar substances dissolve in nonpolar solvents. Miscible liquids mix without separating, while immiscible liquids do not mix.
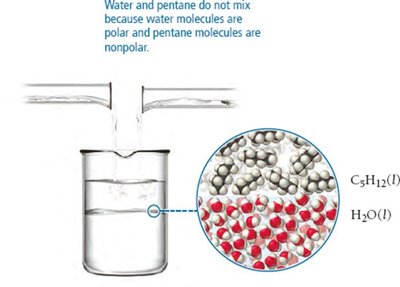
Induced Dipole Interactions
Polar molecules can induce a dipole in nonpolar molecules, allowing them to mix. For example, water (polar) can induce a dipole in O2 (nonpolar), enabling oxygen to dissolve in water.
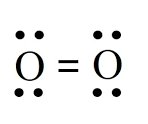
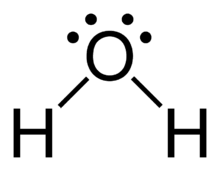
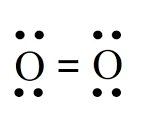

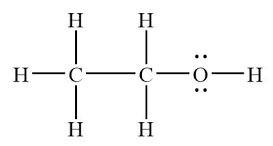
Dipole-Induced Dipole in Ethanol and Iodine
Polar ethanol can induce a dipole in nonpolar I2, allowing I2 to dissolve in ethanol.


Properties Associated with the Liquid State
Viscosity, Surface Tension, and Capillary Action
IMFs influence several properties of liquids:
Viscosity: Resistance to flow; increases with stronger IMFs and higher molar mass, decreases with temperature.
Surface Tension: Tendency to minimize surface area; stronger IMFs lead to higher surface tension.
Capillary Action: Movement of liquid up a thin tube due to adhesive and cohesive forces.
Additional info:
These notes cover the essential concepts of intermolecular forces, their types, and their effects on the physical properties and solubility of substances. The included images directly illustrate molecular arrangements, types of IMFs, and their impact on solubility and physical properties, reinforcing the textual explanations.
