 Back
BackIntermolecular Forces, Bonding, and Properties of Liquids and Solutions
Study Guide - Smart Notes

Bonding and Intermolecular Forces
Covalent, Polar Covalent, and Ionic Bonds
Chemical bonds are classified based on the sharing or transfer of electrons between atoms. The nature of the bond depends on the difference in electronegativity between the atoms involved.
Symmetrical Covalent Bond: Electrons are shared equally between two identical atoms, resulting in a nonpolar bond.
Polar Covalent Bond: Electrons are shared unequally due to differences in electronegativity, creating partial positive (δ+) and partial negative (δ−) charges.
Ionic Bond: Electrons are transferred from one atom to another, forming cations and anions with full charges.
Electronegativity Difference (ΔEN): The degree of bond polarity increases with ΔEN. Bonds are considered "mostly ionic" if ΔEN > 1.7.
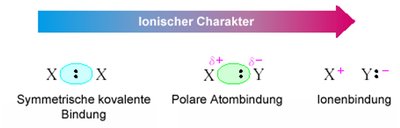
Additional info: The transition from covalent to ionic is gradual, not abrupt.
Electronegativity Trends in the Periodic Table
Electronegativity is a measure of an atom's ability to attract electrons in a bond. It varies across the periodic table.
Highest Electronegativity: Fluorine (F)
Next Highest: Oxygen (O), then Nitrogen (N), closely followed by Chlorine (Cl)
Metals: Generally have low electronegativity
Hydrogen: Intermediate electronegativity
Noble Gases: Typically do not participate in bonding due to full valence shells
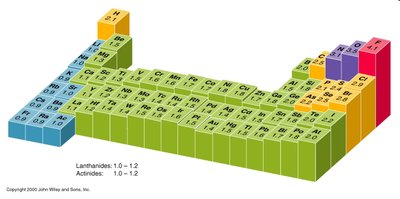
Mnemonic: FONCl (Phone Call) for remembering the order: Fluorine, Oxygen, Nitrogen, Chlorine.
Polarity and Molecular Structure
Polarity of Covalent Bonds
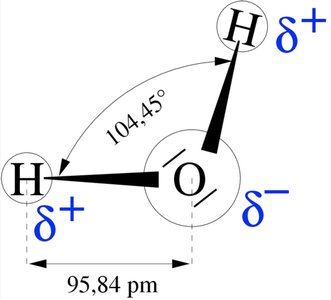
Polarity arises when there is an unequal sharing of electrons, resulting in partial charges. This is especially important in molecules like water.
Water Molecule: The bent structure and high electronegativity of oxygen create a polar molecule with a partial negative charge on oxygen and partial positive charges on hydrogen.
Types of Intermolecular Forces
Overview of Intermolecular Forces
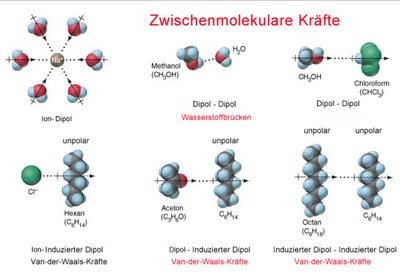
Intermolecular forces are attractions between molecules, influencing physical properties such as boiling point, melting point, and solubility.
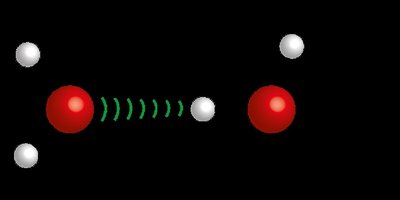
Hydrogen Bonds: Strong dipole-dipole interactions involving hydrogen bonded to N, O, or F.
Dipole-Dipole Interactions: Occur between polar molecules.
Ion-Dipole Interactions: Occur between ions and polar molecules.
Ion-Induced Dipole: An ion induces a dipole in a nearby nonpolar molecule.
London (Van der Waals) Forces: Weak interactions due to temporary dipoles in nonpolar molecules.
Relative Strength of Interactions
The strength of intermolecular forces varies significantly:
Type of Interaction | Dissociation Energy (kJ/mol) |
|---|---|
Covalent (including disulfide bonds) | ~400 |
Hydrogen Bonds | ~20 |
Dipole-Dipole | 0.5 - 2 |
London (Van der Waals) Forces | ~1 |
Additional info: Covalent bonds are much stronger than intermolecular forces.
Hydrogen Bonding in Biomolecules
Hydrogen bonds play a crucial role in the structure of biological molecules, such as DNA and proteins.
DNA: Hydrogen bonds between base pairs stabilize the double helix.
Proteins: Hydrogen bonds contribute to secondary and tertiary structure.

Macroscopic Properties Influenced by Intermolecular Forces
Boiling Points and Molar Mass
Boiling points of substances are influenced by the strength of intermolecular forces and molecular mass.
Hydrogen Bonding: Substances with hydrogen bonds (e.g., H2O, HF) have higher boiling points than expected based on molecular mass.
London Forces: Increase with molecular mass, affecting boiling points of nonpolar molecules.

Surface Tension and Capillary Effect
Surface tension is the result of cohesive forces between molecules at the surface of a liquid. The capillary effect occurs when adhesive forces between a liquid and a solid (e.g., water and glass) draw the liquid up a narrow tube.
Cohesion: Attraction between like molecules (e.g., water-water).
Adhesion: Attraction between unlike molecules (e.g., water-glass).
Meniscus: The curved surface of a liquid in a container, shaped by adhesion and cohesion.


Solubility and "Like Dissolves Like" Principle
Solubility and Intermolecular Forces
The solubility of substances depends on the similarity of intermolecular forces between solute and solvent.
Hydrophilic (water-loving): Substances with hydrogen bonds or polar interactions dissolve well in water.
Lipophilic (fat-loving): Substances with nonpolar interactions (London forces) dissolve well in nonpolar solvents.
Hydrophilic = Lipophobic: Polar substances do not dissolve well in nonpolar solvents.
Lipophilic = Hydrophobic: Nonpolar substances do not dissolve well in water.

Polar Protic and Aprotic Solvents
Polar solvents are classified as protic or aprotic based on their ability to donate protons (hydrogen ions).
Polar Protic: Can form hydrogen bonds and donate protons (e.g., water, alcohols).
Polar Aprotic: Cannot donate protons but are still polar (e.g., DMSO).
Additional info: This classification is important in acid/base chemistry and organic reactions.
Ions and Ionic Compounds
Valence Electrons and Ionic Bond Formation
Valence electrons are the outermost electrons available for bonding. Ionic bonds form when electrons are transferred from one atom to another.
Cations: Positively charged ions formed by loss of electrons (smaller than parent atom).
Anions: Negatively charged ions formed by gain of electrons (larger than parent atom).
Crystal Lattice: Ionic compounds form a regular arrangement of ions in a solid.

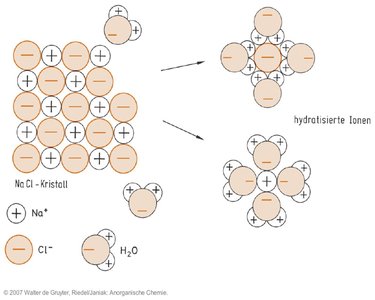
Hydration of Ions
When ionic compounds dissolve in water, ions become surrounded by water molecules, forming hydrated ions.
Hydration: Stabilizes ions in solution and is essential for electrolyte behavior.

Nomenclature of Ionic Compounds
Simple ionic compounds are named by stating the cation first, followed by the anion.
Sodium chloride: NaCl (Na+ and Cl-)
Silver nitrate: AgNO3 (Ag+ and NO3-)
Ammonium bromide: NH4Br (NH4+ and Br-)
Additional info: Molecular ions (e.g., NO3-, NH4+) are common in ionic compounds.
Summary Table: Types of Intermolecular Forces
Type | Example | Relative Strength |
|---|---|---|
Hydrogen Bond | H2O, DNA base pairs | Strong (20 kJ/mol) |
Dipole-Dipole | HCl, acetone | Moderate (0.5-2 kJ/mol) |
Ion-Dipole | Na+ in water | Strong |
London Forces | Hexane, octane | Weak (~1 kJ/mol) |
