 Back
BackIntermolecular Forces, Phase Changes, and Energy Calculations – General Chemistry Study Guide
Study Guide - Smart Notes

Q1. For each of the following compounds, list the intermolecular forces (i.e., dispersion forces, dipole-dipole forces, hydrogen bonding) that they will have with other molecules of the same compound. Which force is the strongest for each substance?
Background
Topic: Intermolecular Forces
This question tests your understanding of the types of intermolecular forces present in different molecular compounds and how to determine which is strongest for each.
Key Terms:
Dispersion forces (London forces): Present in all molecules, especially nonpolar ones.
Dipole-dipole forces: Present in polar molecules.
Hydrogen bonding: A special, strong dipole-dipole interaction when H is bonded to N, O, or F.
Step-by-Step Guidance
For each compound, determine if it is polar or nonpolar by considering its molecular geometry and electronegativity differences.
Identify if the molecule contains H bonded to N, O, or F (for hydrogen bonding).
List all intermolecular forces present for each compound (dispersion is always present).
Decide which force is the strongest for each compound based on its structure and bonding.
Try solving on your own before revealing the answer!
Q2. For each of the following, identify the intermolecular forces between the compound and i) water; ii) hexane (C6H14).
Background
Topic: Intermolecular Forces in Mixtures
This question asks you to consider how different compounds interact with water (polar, hydrogen bonding) and hexane (nonpolar, dispersion forces).
Key Terms:
Water: Polar, hydrogen bonding.
Hexane: Nonpolar, only dispersion forces.
Step-by-Step Guidance
For each compound, determine its polarity and ability to hydrogen bond.
Consider how it would interact with water (look for hydrogen bonding and dipole-dipole interactions).
Consider how it would interact with hexane (primarily dispersion forces).
List the types of intermolecular forces for each pair.
Try solving on your own before revealing the answer!
Q3. Rank the noble gases in order from lowest to highest boiling point.
Background
Topic: Trends in Boiling Points
This question tests your understanding of how atomic size and dispersion forces affect boiling points in noble gases.
Key Terms:
Boiling point: Temperature at which a substance changes from liquid to gas.
Dispersion forces: Increase with larger, more polarizable electron clouds.
Step-by-Step Guidance
List the noble gases (He, Ne, Ar, Kr, Xe, Rn).
Recall that boiling point increases with atomic mass and size due to stronger dispersion forces.
Arrange the noble gases from smallest to largest atomic number.
Rank them from lowest to highest boiling point based on their size.
Try solving on your own before revealing the answer!
Q4. Rank the following from lowest to highest boiling point: MgCl2, H2O, O2, CH2F2. Explain your reasoning.
Background
Topic: Boiling Point and Intermolecular Forces
This question asks you to compare substances with different types of intermolecular forces and predict their boiling points.
Key Terms:
Ionic compounds: Very high boiling points (MgCl2).
Hydrogen bonding: High boiling points (H2O).
Dipole-dipole: Moderate boiling points (CH2F2).
Dispersion forces: Lowest boiling points (O2).
Step-by-Step Guidance
Identify the type of intermolecular force for each compound.
Recall the general trend: ionic > hydrogen bonding > dipole-dipole > dispersion.
Arrange the compounds in order of increasing boiling point.
Explain your reasoning based on the forces present.
Try solving on your own before revealing the answer!
Q5. Use the table to calculate how much energy (in kJ) is required to convert:
3.25 mol of ethanol from liquid to gas at 78.3 °C
0.342 mol of benzene from liquid to gas at 80.1 °C
3.25 mol of ethanol from solid to liquid at -117.3 °C
0.342 mol of benzene from solid to liquid at 5.5 °C
25.0 g of H2O from ice to water at 0 °C
25.0 g of H2O from water to steam at 100 °C
Background
Topic: Phase Changes and Enthalpy
This question tests your ability to use enthalpy of fusion () and enthalpy of vaporization () to calculate energy required for phase changes.
Key Formulas:
Where is energy (kJ), is moles, is enthalpy change (kJ/mol).
For mass:
Step-by-Step Guidance
Identify the phase change (fusion or vaporization) and the relevant value from the table.
For each part, determine the number of moles (if given mass, convert to moles).
Multiply the number of moles by the appropriate value to find the energy required.
Set up the calculation for each part, but do not compute the final value yet.
Try solving on your own before revealing the answer!
Q6. How much energy (in kJ) is required to convert 32.0 g of water at 45 °C to steam at 123 °C? (specific heat of water: 4.18 J/g·°C, specific heat of steam: 1.99 J/g·°C)
Background
Topic: Heating Curves and Energy Calculations
This question tests your ability to calculate total energy for temperature changes and phase changes using specific heat and enthalpy values.
Key Formulas:
(for temperature change)
(for vaporization)
Sum the energy for heating liquid, vaporizing, and heating steam.
Step-by-Step Guidance
Calculate energy to heat water from 45 °C to 100 °C using .
Calculate energy to vaporize water at 100 °C using .
Calculate energy to heat steam from 100 °C to 123 °C using .
Add all energy values for total energy required.
Try solving on your own before revealing the answer!
Q7. Answer the questions based on the following heating curve for a substance:
a. Identify what is happening on a molecular level at each of segments 1-5
b. Why is the horizontal line at 4 longer than the horizontal line at 2?
c. Why would the line at 5 be steeper than the line at 3?
Background
Topic: Heating Curves and Phase Changes
This question tests your understanding of the molecular events during heating, melting, and boiling, and how energy is absorbed during phase changes.
Key Terms:
Heating curve: Graph showing temperature change as a substance is heated.
Plateau (horizontal line): Phase change (melting or boiling), temperature remains constant.
Slope (steepness): Related to specific heat capacity.
Step-by-Step Guidance
For each segment, describe whether the substance is being heated (solid, liquid, gas) or undergoing a phase change (melting, boiling).
Explain why the length of the plateau (horizontal line) is related to the amount of energy required for the phase change.
Discuss why the slope of the line depends on the specific heat capacity of the phase.
Relate these concepts to the molecular behavior (breaking intermolecular forces, increasing kinetic energy).
Try solving on your own before revealing the answer!
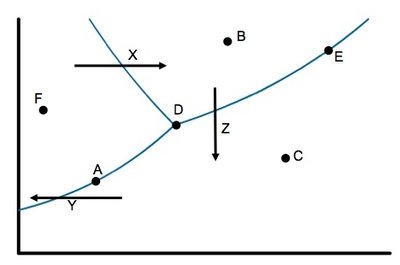
Q8. Using the phase diagram on the following page:
a. Identify the states present at points A-F
b. Identify the phase changes indicated by arrows X, Y, and Z.
Background
Topic: Phase Diagrams
This question tests your ability to interpret phase diagrams and identify the physical states and phase transitions at different points and along different paths.
Key Terms:
Phase diagram: Graph showing regions of solid, liquid, and gas as a function of temperature and pressure.
Triple point: Point where all three phases coexist.
Critical point: End of the liquid-gas boundary.
Step-by-Step Guidance
Examine the phase diagram and locate points A-F. Determine which region (solid, liquid, gas) each point is in based on its position relative to the phase boundaries.
For each arrow (X, Y, Z), identify the direction of movement and which phase change it represents (e.g., melting, vaporization, sublimation).
Use the diagram to match each point and arrow to the correct phase or transition.
Try solving on your own before revealing the answer!
