 Back
BackIntermolecular Forces: Types, Effects, and Examples
Study Guide - Smart Notes

Intermolecular Forces (IMFs)
Definition and Importance
Intermolecular forces (IMFs) are the attractions and repulsions between atoms and molecules that determine the physical properties of substances, such as melting point, boiling point, solubility, and the existence of solids and liquids. IMFs are much weaker than chemical bonds but are crucial for understanding condensed phases of matter.
Comparison of Chemical Bonds and IMFs
Chemical bonds (e.g., covalent bonds) are strong and hold atoms together within molecules.
IMFs are weaker and act between molecules, influencing their physical behavior.
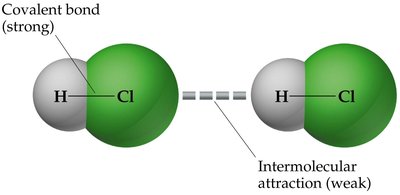

Types of Intermolecular Forces
Overview
The strength and type of IMFs depend on three main properties:
Presence of charge (ion or not)
Polarity (molecular shape, dipoles)
Molar mass (size of molecule)
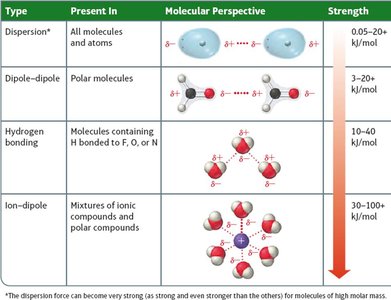
Type | Present In | Molecular Perspective | Strength |
|---|---|---|---|
Dispersion* | All molecules and atoms |
| 0.05–20 kJ/mol |
Dipole–Dipole | Polar molecules |
| 3–20 kJ/mol |
Hydrogen Bonding | Molecules containing H bonded to F, O, or N |
| 10–40 kJ/mol |
Ion–Dipole | Mixtures of ionic compounds and polar compounds |
| 30–100+ kJ/mol |
Ion-Dipole Forces
Ion-dipole forces occur when ions from an ionic compound interact with the dipole of polar molecules. This is a key factor in the solubility of ionic compounds in water.
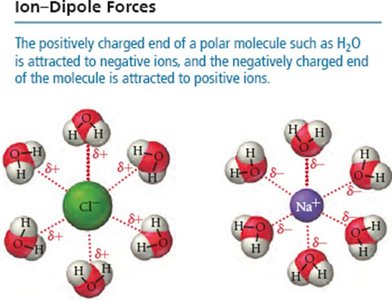
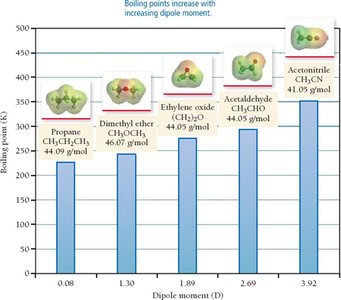
Dipole-Dipole Forces
Dipole-dipole forces exist between polar molecules with permanent dipoles. The positive end of one molecule is attracted to the negative end of another, increasing boiling and melting points compared to nonpolar molecules of similar size.
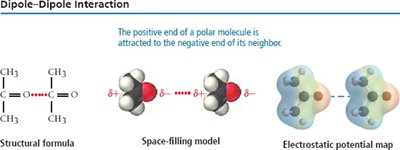

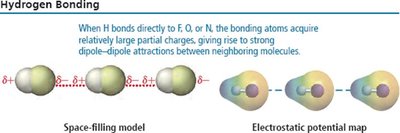
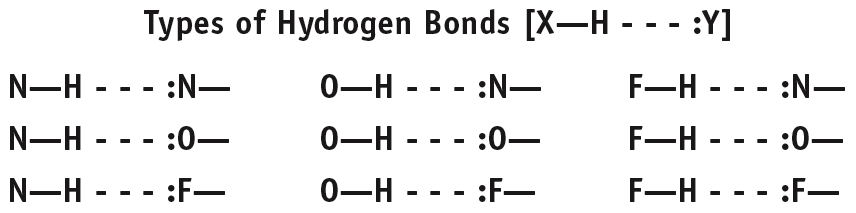
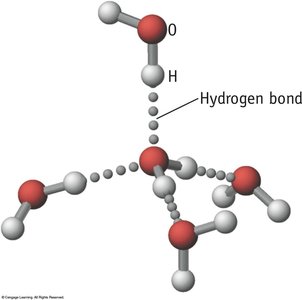
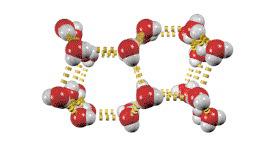
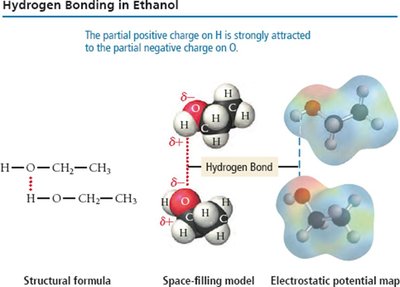
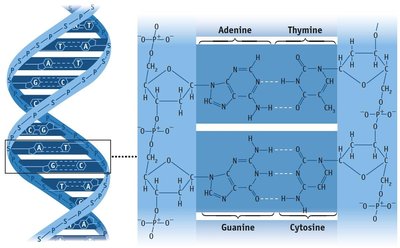
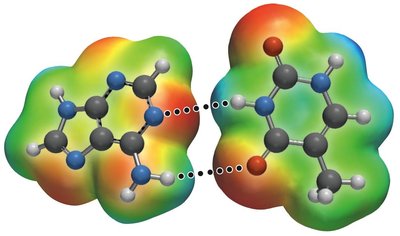
Hydrogen Bonding
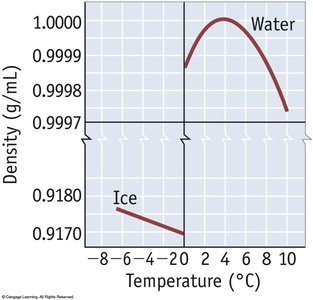
Hydrogen bonding is a particularly strong type of dipole-dipole interaction, occurring when hydrogen is bonded to N, O, or F. It significantly affects properties such as boiling point, melting point, and the structure of water and biological molecules.
Hydrogen bonds are not as strong as covalent bonds but are stronger than other IMFs.
Substances with hydrogen bonding have higher boiling and melting points.
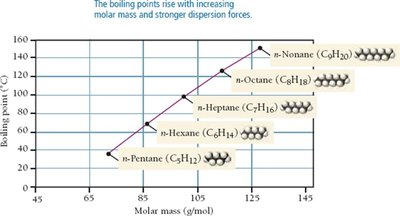
Dispersion Forces (London Forces)
Dispersion forces arise from temporary fluctuations in electron distribution, creating instantaneous dipoles. These forces are present in all molecules and atoms, but are the only IMFs in nonpolar molecules.
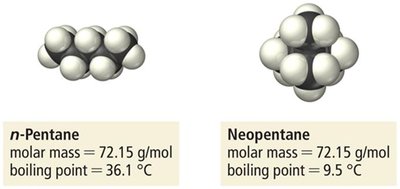
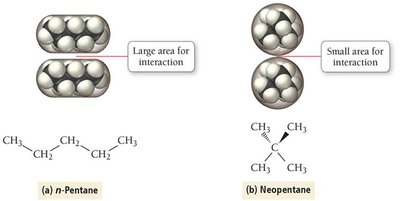
Strength increases with molar mass and polarizability.
Molecular shape affects the magnitude: straight chains have higher boiling points than branched chains due to greater surface contact.

Summary Table: Intermolecular Forces
Type | Interacting Particles | Polar Molecules? | Ions? | Hydrogen Bonding? |
|---|---|---|---|---|
London Forces | All molecules/atoms | No | No | No |
Dipole–Dipole | Polar molecules | Yes | No | No |
Hydrogen Bonding | H bonded to N, O, F | Yes | No | Yes |
Ion–Dipole | Ions and polar molecules | Yes | Yes | No |
Ionic Bonding | Ions | No | Yes | No |
Effects of IMFs on Physical Properties
Boiling and Melting Points
The strength of IMFs directly affects boiling and melting points. Stronger IMFs result in higher boiling and melting points for substances of similar molar mass.
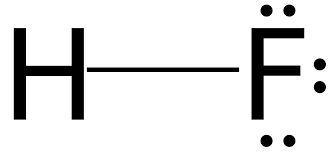
Solubility and Miscibility
IMFs also determine solubility and miscibility. "Like dissolves like": polar substances dissolve in polar solvents, nonpolar substances in nonpolar solvents. Molecules with both hydrophilic and hydrophobic parts may have complex solubility behavior.
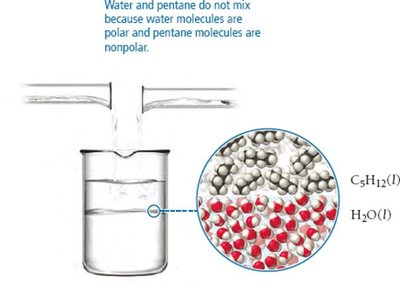
Induced Dipole Forces
Polar molecules can induce dipoles in nonpolar molecules, allowing them to mix. For example, water (polar) can induce a dipole in O2 (nonpolar), enabling oxygen to dissolve in water.
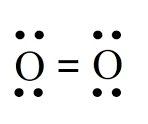
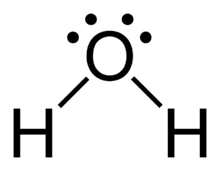
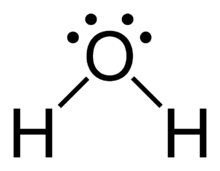




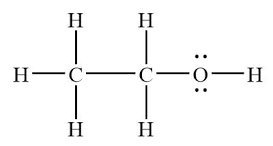
Key Takeaways
IMFs are essential for understanding the physical properties of substances.
There are several types of IMFs: dispersion, dipole-dipole, hydrogen bonding, and ion-dipole.
IMFs influence boiling/melting points, solubility, and miscibility.
Polar molecules can induce dipoles in nonpolar molecules, enabling mixing.
