 Back
BackIntermolecular Forces: Types, Strengths, and Effects on Properties
Study Guide - Smart Notes

Intermolecular Forces (IMFs)
Definition and Importance
Intermolecular forces (IMFs) are the attractions and repulsions between atoms and molecules that determine the physical properties of substances, such as melting point, boiling point, solubility, and phase behavior. IMFs are much weaker than chemical bonds but are crucial for the existence of condensed phases (liquids and solids).
IMFs vs. Chemical Bonds: IMFs are weaker than covalent or ionic bonds, but they significantly influence physical properties.
Example: The energy required to break an H–Cl covalent bond is much higher than the energy needed to overcome IMFs between HCl molecules.
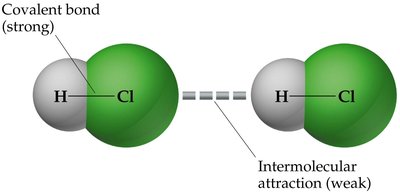

Types of Intermolecular Forces
Classification and Molecular Properties
The type and strength of IMFs depend on three main molecular properties:
Charge: Presence of ions leads to ion-dipole interactions.
Polarity: Polar molecules exhibit dipole-dipole and hydrogen bonding.
Molar Mass: Larger molecules have stronger London dispersion forces.
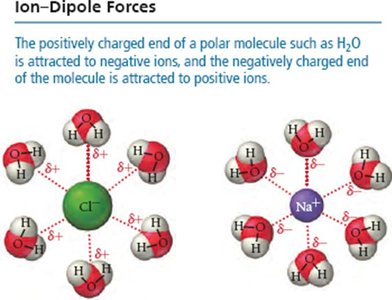
Ion-Dipole Forces
Ion-dipole forces occur between ions and polar molecules. These are especially important in solutions of ionic compounds in polar solvents like water.
Strength: Among the strongest IMFs, crucial for solubility of salts in water.
Example: Na+ and Cl– ions surrounded by water molecules.
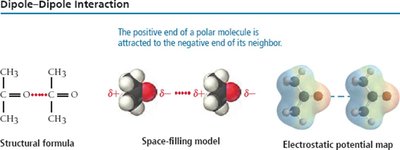
Dipole-Dipole Forces
Dipole-dipole forces occur between polar molecules, where the positive end of one molecule is attracted to the negative end of another.
Strength: Moderate, increases with molecular polarity.
Effect: Raises boiling and melting points compared to nonpolar molecules of similar size.
Example: Formaldehyde (CH2O) vs. Ethane (C2H6).

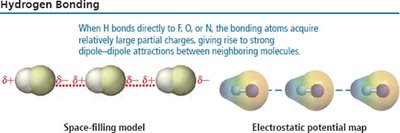
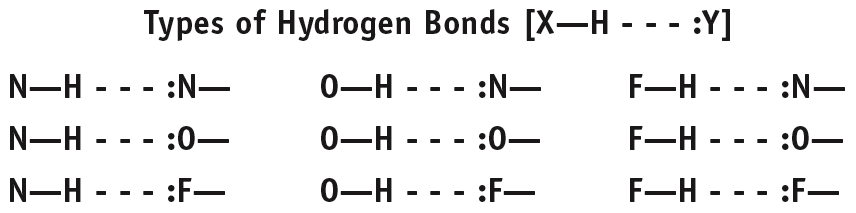
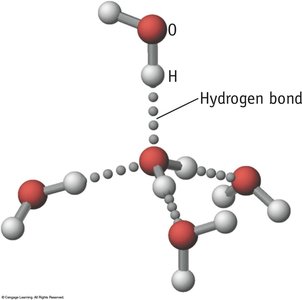
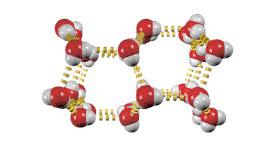
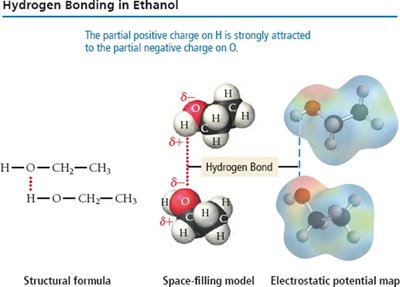
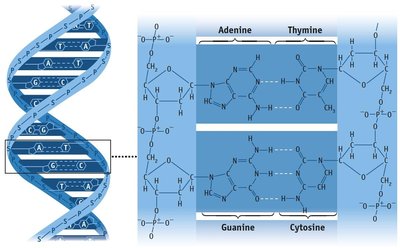
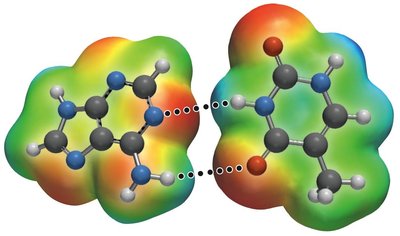
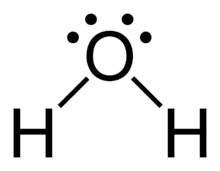
Hydrogen Bonding
Hydrogen bonding is a particularly strong type of dipole-dipole interaction, occurring when hydrogen is bonded to highly electronegative atoms (N, O, or F).
Strength: Strongest among dipole-dipole forces, but weaker than covalent bonds.
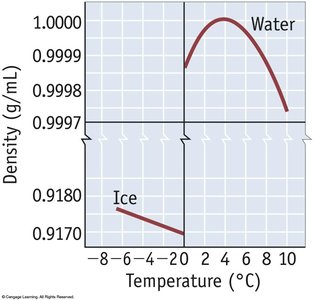
Effect: Significantly increases boiling and melting points; responsible for unique properties of water.
Examples: Water, ethanol, DNA base pairing.
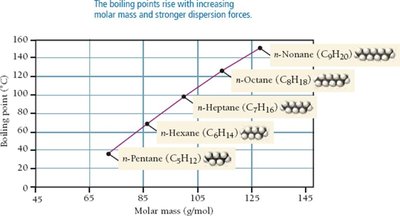
London Dispersion Forces (Van der Waals Forces)
Dispersion forces arise from temporary fluctuations in electron distribution, creating instantaneous dipoles that induce dipoles in neighboring molecules. These are present in all molecules, but are the only IMFs in nonpolar substances.
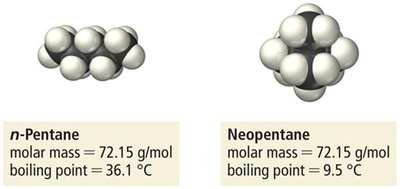
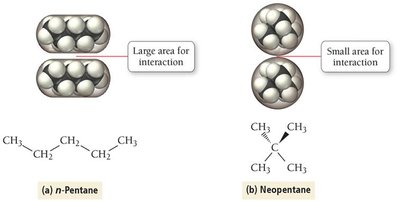
Strength: Weakest, but increases with molar mass and molecular size.
Effect: Responsible for boiling point trends in noble gases and hydrocarbons.
Example: n-Pentane vs. Neopentane; boiling points increase with surface area and molar mass.

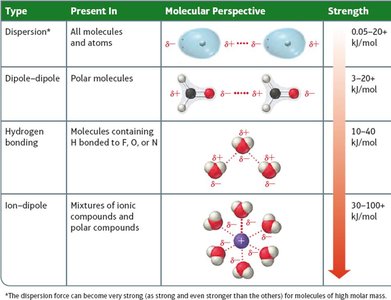
Summary Table: Types of Intermolecular Forces
Type | Present In | Molecular Perspective | Strength |
|---|---|---|---|
Dispersion | All molecules and atoms | Temporary dipoles | 0.05–20 kJ/mol |
Dipole–Dipole | Polar molecules | Permanent dipoles | 3–20 kJ/mol |
Hydrogen Bonding | Molecules with H bonded to F, O, or N | Strong dipole–dipole | 10–40 kJ/mol |
Ion–Dipole | Mixtures of ions and polar compounds | Ion and dipole interaction | 30–100+ kJ/mol |
Effects of IMFs on Physical Properties
Boiling and Melting Points
The strength and type of IMFs directly affect boiling and melting points. Stronger IMFs result in higher boiling and melting points.
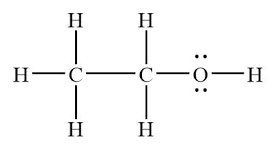
Example: Ethanol (hydrogen bonding) has a higher boiling point than diethyl ether (dipole-dipole and dispersion only).
Solubility
IMFs determine solubility: "like dissolves like." Polar substances dissolve in polar solvents, nonpolar in nonpolar solvents. Ion-dipole forces are key for dissolving salts in water.
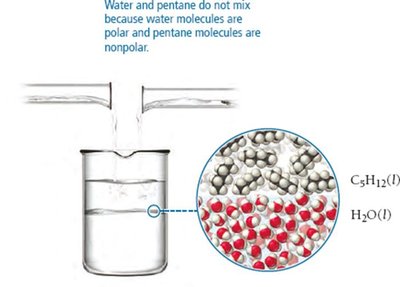
Induced Dipole Forces
Polar molecules can induce dipoles in nonpolar molecules, allowing some mixing (e.g., O2 in water, I2 in ethanol).


Summary
IMFs are essential for understanding the physical properties of substances.
Types of IMFs: Ion-dipole, dipole-dipole, hydrogen bonding, and London dispersion.
Strength order: Ion-dipole > Hydrogen bonding > Dipole-dipole > Dispersion.
IMFs affect boiling/melting points, solubility, and phase behavior.
