 Back
BackIntroduction to General Chemistry: Matter, Properties, and Measurement
Study Guide - Smart Notes

Introduction to Chemistry
Chemistry as a Science
Chemistry is the science that seeks to understand the behavior of matter and the changes it undergoes. Matter is defined as anything that has mass and occupies space. Chemistry explores why substances behave differently under various conditions, such as why fire melts ice but burns paper.
The Scientific Method
The scientific method is the systematic approach scientists use to study the world. - Observation: Gathering information about phenomena. - Hypothesis: A tentative explanation for observations. - Experiment: Controlled procedures to test hypotheses. - Law: A statement summarizing repeated observations and predicting future ones. - Theory: An explanation that accounts for laws and observations, often predicting behavior beyond initial findings. Key distinction: A law summarizes what happens; a theory explains why it happens.
Classification of Matter
States of Matter
Matter exists in three primary states: solid, liquid, and gas. - Solid: Atoms or molecules are fixed in place and can only vibrate. - Liquid: Atoms or molecules are closely packed but can move past one another, allowing the liquid to flow. - Gas: Atoms or molecules are widely spaced and move freely, making gases compressible and able to flow. 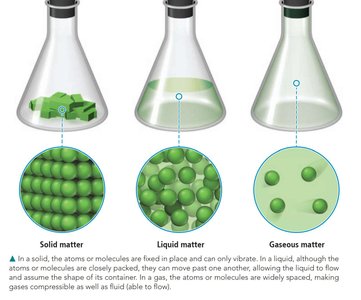
Classification by Composition
Matter can be classified as pure substances or mixtures. - Pure Substance: Composed of one component with invariant composition. - Element: Cannot be chemically broken down into simpler substances. - Compound: Composed of two or more elements in fixed proportions. - Mixture: Composed of two or more components in variable proportions. - Heterogeneous Mixture: Composition varies from one region to another. - Homogeneous Mixture: Uniform composition throughout. 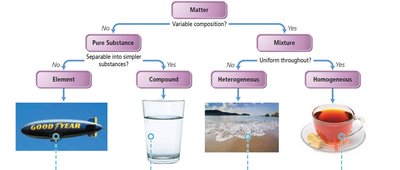
Microscopic Representation of Matter
Visual models help distinguish between elements, compounds, and mixtures at the atomic level. 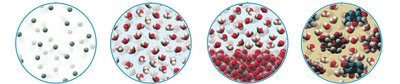
Separation of Mixtures
Physical Separation Techniques
Mixtures can be separated by physical methods such as distillation and filtration. - Distillation: Separates components based on differences in boiling points. - Filtration: Separates solids from liquids using filter paper. 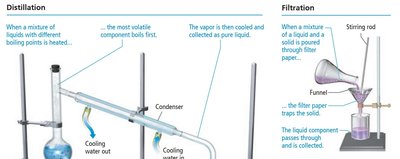

Physical and Chemical Changes
Physical Changes
Physical changes alter only the state or appearance of matter, not its composition. Examples include melting, boiling, and sublimation. 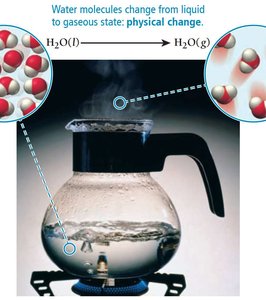

Chemical Changes
Chemical changes alter the composition of matter, resulting in the formation of new substances. Examples include burning, rusting, and bleaching. 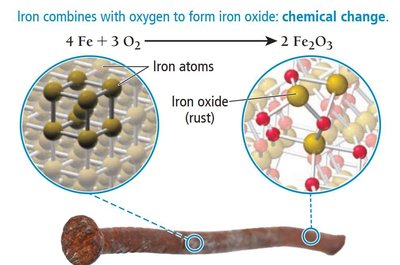
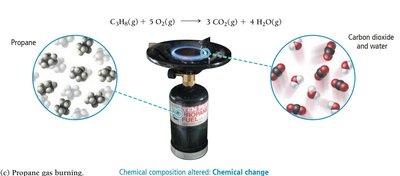
Properties of Matter
- Physical Property: Observed without changing composition (e.g., melting point, density). - Chemical Property: Observed only by changing composition (e.g., flammability, acidity).
Energy in Chemistry
Types of Energy
Energy is the capacity to do work. - Kinetic Energy: Energy of motion. - Potential Energy: Energy due to position. - Thermal Energy: Energy associated with temperature, a form of kinetic energy.  Conservation of Energy: Energy is always conserved in physical and chemical changes; it is neither created nor destroyed.
Conservation of Energy: Energy is always conserved in physical and chemical changes; it is neither created nor destroyed.
Measurement in Chemistry
Measurement and Units
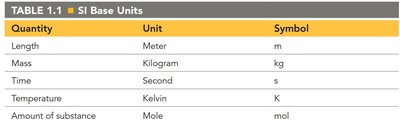
Measurements consist of a number and a unit. Chemistry uses the SI (International System) units for standardization.
Quantity | Unit | Symbol |
|---|---|---|
Length | Meter | m |
Mass | Kilogram | kg |
Time | Second | s |
Temperature | Kelvin | K |
Amount of substance | Mole | mol |
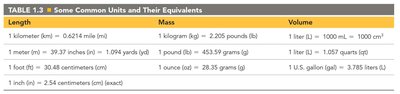
Common Units and Equivalents
Chemistry often uses conversions between units.
Length | Mass | Volume |
|---|---|---|
1 kilometer (km) = 0.6214 mile (mi) | 1 kilogram (kg) = 2.205 pounds (lb) | 1 liter (L) = 1000 mL = 1000 cm3 |
1 meter (m) = 39.37 inches (in) = 1.094 yards (yd) | 1 pound (lb) = 453.59 grams (g) | 1 liter (L) = 1.057 quarts (qt) |
1 foot (ft) = 30.48 centimeters (cm) | 1 ounce (oz) = 28.35 grams (g) | 1 U.S. gallon (gal) = 3.785 liters (L) |
1 inch (in) = 2.54 centimeters (cm) |
Temperature Scales
The SI unit for temperature is the kelvin (K), which measures average kinetic energy. - Kelvin: Absolute scale, no negative values. - Celsius: Based on water's freezing and boiling points. - Fahrenheit: Commonly used in the U.S. Conversion formulas:
SI Prefixes and Derived Units
SI Prefixes
Prefixes are used to represent multiples or fractions of units.  Examples: - 1 kilometer = 1000 meters = meters - 1 millimeter = 0.001 meters = meters
Examples: - 1 kilometer = 1000 meters = meters - 1 millimeter = 0.001 meters = meters
Derived Units: Volume and Density
Some properties require combinations of base units. - Volume: Space occupied, measured in , , or . - Density: Mass per unit volume. Intensive property: Independent of amount (e.g., density). Extensive property: Dependent on amount (e.g., volume).
Example: Calculating Density
If a ring has a mass of 3.15 g and displaces 0.233 cm3 of water, its density is: Comparison: Platinum's density is 21.4 g/cm3, so the ring is not platinum.
Summary Table: Classification of Matter
Type | Description | Example |
|---|---|---|
Element | Cannot be broken down | Oxygen (O2) |
Compound | Two or more elements, fixed ratio | Water (H2O) |
Homogeneous Mixture | Uniform composition | Salt water |
Heterogeneous Mixture | Non-uniform composition | Sand and water |
Additional info: Academic context was added to clarify definitions, provide examples, and expand explanations for completeness.
