 Back
BackIntroduction to Organic Compounds: Structure, Nomenclature, and Isomerism
Study Guide - Smart Notes

Organic Chemistry: Introduction and Overview
Definition and Scope of Organic Chemistry
Organic chemistry is the branch of chemistry that studies compounds primarily composed of carbon and hydrogen, often containing other elements such as oxygen, nitrogen, sulfur, and halogens. Organic compounds are the foundation of all life on Earth and are central to biochemistry, medicine, and many industrial processes.
Organic Compounds: Molecules containing carbon atoms bonded to hydrogen, and often to other elements.
Inorganic Compounds: Compounds not primarily based on carbon-hydrogen frameworks (e.g., KBr, FeCl2).
Examples: CH4 (methane), C6H12O7 (a carbohydrate).
Representing Organic Molecules
Molecular, Condensed, Structural, and Skeletal Formulas
Organic molecules can be represented in several ways, each providing different levels of detail about the connectivity and arrangement of atoms.
Molecular Formula: Shows the number and type of atoms (e.g., C4H10 for butane).
Condensed Formula: Groups atoms to show connectivity (e.g., CH3CH2CH2CH3).
Structural Formula: Shows all bonds between atoms explicitly.
Skeletal Formula: Simplifies the structure by representing carbon atoms as vertices and omitting hydrogen atoms bonded to carbon.
Lewis Structure: Shows all atoms, bonds, and lone pairs.

Hydrocarbons: Classification and Structure
Types of Hydrocarbons
Hydrocarbons are organic compounds consisting entirely of carbon and hydrogen. They are classified based on the types of bonds between carbon atoms.
Alkanes: Only single C–C bonds (saturated hydrocarbons).
Alkenes: Contain at least one C=C double bond (unsaturated hydrocarbons).
Alkynes: Contain at least one C≡C triple bond (unsaturated hydrocarbons).
Aromatic Compounds: Contain planar ring structures with delocalized electrons (e.g., benzene).

Alkanes: Structure and Nomenclature
Alkanes are the simplest hydrocarbons, containing only single bonds. They can be straight-chain or branched.
General Formula: for straight-chain alkanes.
Examples: Methane (CH4), Ethane (C2H6), Propane (C3H8), Butane (C4H10).

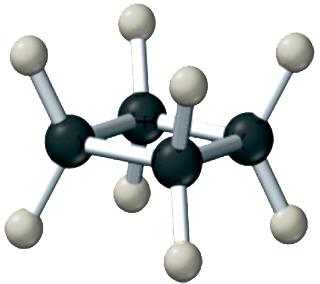
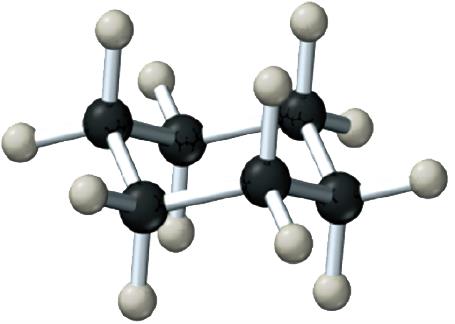
Cycloalkanes
Cycloalkanes are saturated hydrocarbons with carbon atoms arranged in rings. Their general formula is .
Cyclopropane: Triangle ring (C3H6).
Cyclobutane: Square ring (C4H8).
Cyclopentane: Pentagon ring (C5H10).
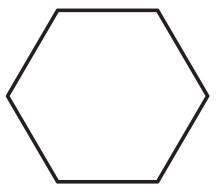
Cyclohexane: Hexagon ring (C6H12).


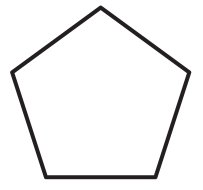
Functional Groups in Organic Chemistry
Definition and Importance
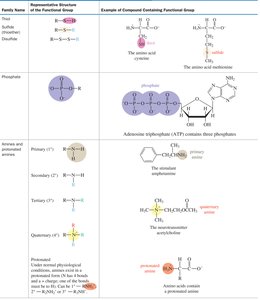
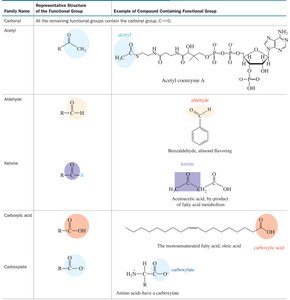
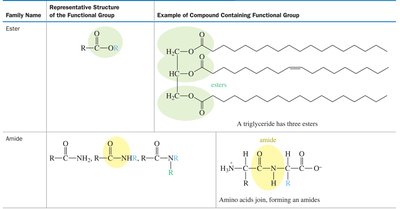
Functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. Recognizing functional groups is essential for understanding organic reactivity and nomenclature.
Examples: Alcohols (–OH), Amines (–NH2), Carboxylic acids (–COOH), Esters (–COOR), Amides (–CONH2).
Reactivity: Functional groups determine the types of chemical reactions a molecule can undergo.
Unsaturated Hydrocarbons
Alkenes and Alkynes
Unsaturated hydrocarbons contain one or more double or triple bonds between carbon atoms, making them more reactive than alkanes.
Alkenes: Contain C=C double bonds. General formula: .
Alkynes: Contain C≡C triple bonds. General formula: .
Bond Reactivity: Double and triple bonds are more reactive and easier to break than single bonds.


Aromatic Compounds
Aromatic compounds are cyclic, planar molecules with delocalized electrons, typically based on the benzene ring. They are unusually stable due to resonance.
Benzene: The prototypical aromatic compound, with alternating double bonds in a six-membered ring.
Stability: Aromatic compounds are more stable than alkenes due to electron delocalization.
Isomerism in Organic Compounds
Structural Isomers
Structural isomers have the same molecular formula but different connectivity of atoms.
Example: n-propyl alcohol and isopropyl alcohol (C3H8O).
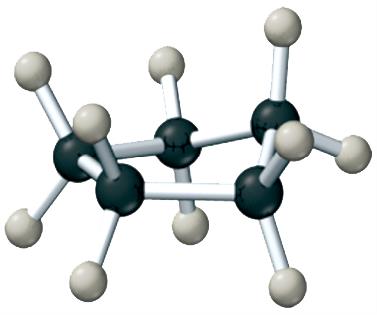
Conformational Isomers
Conformational isomers differ by rotation around single bonds and do not require breaking bonds to interconvert.
Example: Different conformations of butane.
Stereoisomers
Stereoisomers have the same molecular formula and connectivity but differ in the spatial arrangement of atoms.
Cis-Trans Isomerism: Occurs in alkenes and cyclic compounds when substituents are on the same (cis) or opposite (trans) sides.
Enantiomers: Non-superimposable mirror images, often due to the presence of a chiral center (a carbon with four different substituents).
Nomenclature of Organic Compounds
Alkane Naming and IUPAC Rules
Organic compounds are named systematically using the IUPAC system, which identifies the longest carbon chain, names substituents, and assigns numbers to minimize the position numbers of substituents.
Parent Chain: The longest continuous chain of carbon atoms.
Substituents: Groups attached to the parent chain, named as prefixes.
Numbering: The chain is numbered to give the lowest possible numbers to substituents.
Suffix: Indicates the family (e.g., -ane for alkanes, -ene for alkenes).

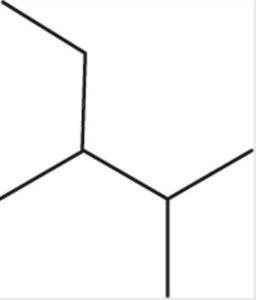
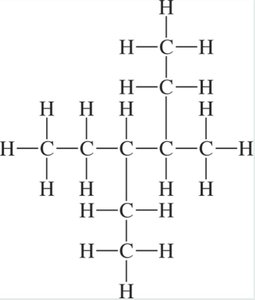
Haloalkanes
Haloalkanes are alkanes with one or more halogen atoms (F, Cl, Br, I) as substituents. The halogen is named as a prefix (e.g., bromo-, chloro-).

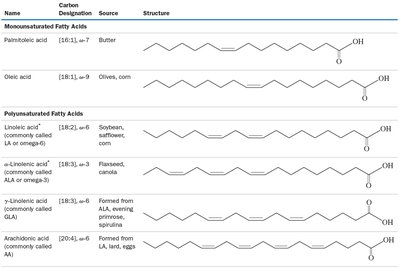
Fatty Acids and Lipids
Saturated and Unsaturated Fatty Acids
Fatty acids are long-chain carboxylic acids found in lipids. They can be saturated (no double bonds), monounsaturated (one double bond), or polyunsaturated (multiple double bonds).
Saturated Fatty Acids: Only single bonds between carbons (e.g., lauric acid, stearic acid).
Unsaturated Fatty Acids: Contain one or more C=C double bonds (e.g., linoleic acid).
Physical Properties: Unsaturated fats are generally more fluid and easier to break down than saturated fats.
Summary Table: Hydrocarbon Families and Functional Groups
Family Member | Functional Group |
|---|---|
Alkane | None; contain only C and H single bonds |
Alkene | C=C double bond |
Alkyne | C≡C triple bond |
Aromatic | Planar, ring structures based on benzene; delocalized electrons |
*Additional info: This guide covers the foundational concepts of organic chemistry relevant to a general chemistry course, including molecular representations, hydrocarbon classification, functional groups, isomerism, and nomenclature. The included images directly support the understanding of molecular structures, functional groups, and naming conventions.*
