 Back
BackIntroduction to Solutions and Aqueous Reactions: Study Guide
Study Guide - Smart Notes

Introduction to Solutions and Aqueous Reactions
Basic Definitions and Concepts
Solutions are fundamental in chemistry, especially in aqueous reactions where water acts as the solvent. Understanding the terminology and properties of solutions is essential for studying chemical reactions in water.
Solution: A homogeneous mixture of two or more substances. Example: salt dissolved in water.
Solvent: The component present in the highest proportion in a solution; in aqueous solutions, this is water.
Solute: The minority component(s) in the mixture; the substance(s) dissolved in the solvent.
Aqueous solution: A solution in which water is the solvent.



Identifying Solvent and Solute
In mixtures, the solvent is the substance in the greatest amount, while the solute(s) are present in lesser amounts. Sometimes, mixtures can have more than one solvent or solute.
Example 1: 35% naphthalene and 65% benzene: Benzene is the solvent, naphthalene is the solute.
Example 2: 10% ethanol, 40% methanol, 50% propanol: Propanol is the solvent, ethanol and methanol are solutes.
Example 3: 40% ethanol, 40% methanol, 20% butanol: Ethanol and methanol are mixed solvents, butanol is the solute.
Example 4: 40% ethanol, 50% propanol, 10% water: Ethanol and propanol are solvents, water is the solute.
Solution Concentration
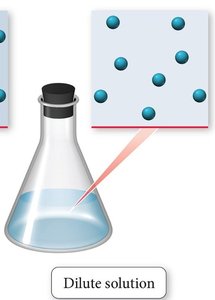
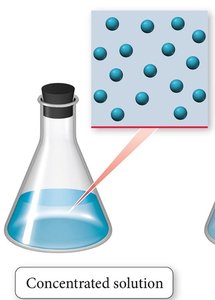
Dilute vs. Concentrated Solutions
Solutions are often described as dilute or concentrated based on the relative amount of solute compared to solvent.
Dilute solution: Contains a small amount of solute relative to the solvent.
Concentrated solution: Contains a large amount of solute relative to the solvent.
Quantifying Solution Concentration: Molarity
Molarity (M) is the most common way to express solution concentration in chemistry. It is defined as the number of moles of solute per liter of solution.
Formula: where n = moles of solute, V = volume of solution in liters.
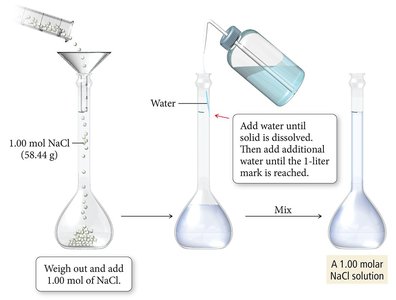
Preparing a 1.00 M NaCl Solution
To prepare a 1.00 M solution of sodium chloride (NaCl), weigh out 1.00 mol (58.44 g) of NaCl, dissolve it in water, and dilute to a final volume of 1 liter.
Calculating Molarity: Example Problems
Example 1: What is the molarity of a solution containing 25.5 g KBr dissolved in enough water to make 1.75 L solution?
Calculate moles of KBr:
Calculate molarity:
Example 2: What mass of KBr is needed to make 250.0 mL of a 1.50 M KBr solution?
Convert volume to liters:
Calculate moles:
Calculate mass:
Solution Dilution
Concept of Dilution
Stock solutions are often concentrated and must be diluted to obtain desired concentrations. Dilution involves adding more solvent, which increases the volume but keeps the amount of solute constant.
Key equation: where M = molarity, V = volume, subscripts 1 and 2 refer to initial and final conditions.

Example: Diluting a Stock Solution
To prepare 3.00 L of 0.500 M CaCl2 from a 10.0 M stock solution:
Example: Calculating Final Volume After Dilution
To dilute 100.0 mL of 5.00 M CaCl2 to obtain a 0.750 M solution:
Solvent to add:
Solution Stoichiometry
Stoichiometric Calculations in Solution
Stoichiometry in solutions involves using molarity, volume, and balanced chemical equations to calculate the amounts of reactants and products.
General reaction:
Key steps:
Calculate moles of reactant using
Use mole ratios from the balanced equation to find moles of other substances
Convert moles back to volume if needed using
Types of Aqueous Solutions and Solubility
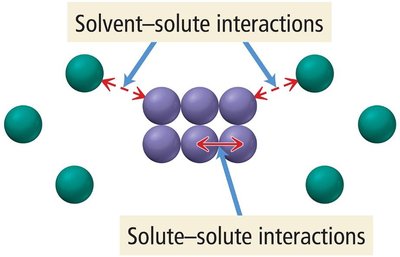
Solubility and Dissolution
Solubility describes how well a solute dissolves in a solvent. When a solute dissolves, attractive forces between solute and solvent must overcome solute-solute and solvent-solvent interactions.
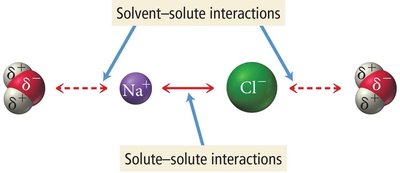
Dissolution of Ionic Compounds
When ionic compounds dissolve in water, ions are separated and surrounded by water molecules, resulting in free-moving charged particles that can conduct electricity.
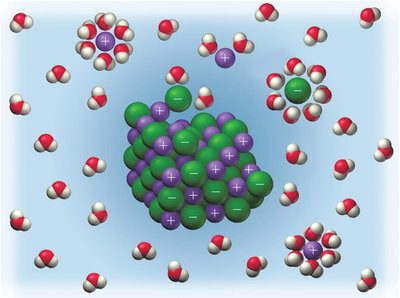
Electrolyte and Nonelectrolyte Solutions
Electrolytes are substances that dissolve in water to produce ions and conduct electricity. Nonelectrolytes dissolve without forming ions and do not conduct electricity.
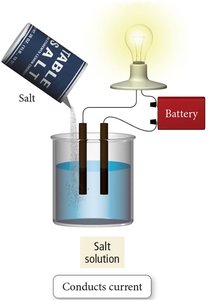
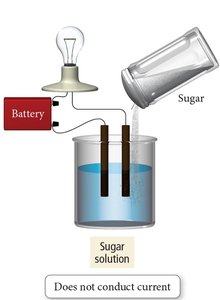
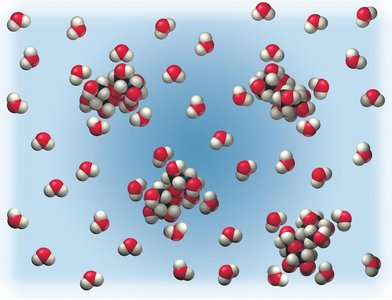
Salt vs. Sugar Dissolved in Water
Ionic compounds like NaCl dissociate into ions in water and are strong electrolytes. Molecular compounds like sugar (C6H12O6) do not dissociate and are nonelectrolytes.
Strong and Weak Electrolytes
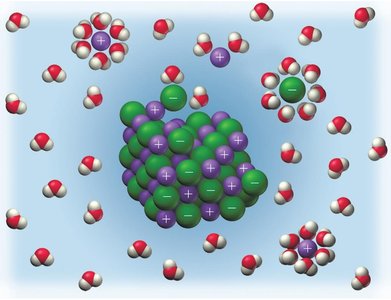
Strong electrolytes dissociate completely in water, while weak electrolytes only partially dissociate. Strong acids and bases are strong electrolytes; weak acids are weak electrolytes.
Dissociation and Ionization
Dissociation of Ionic Compounds
When ionic compounds dissolve, their ions separate. Polyatomic ions remain intact during dissociation. Strong acids ionize completely in water.
Solubility of Ionic Compounds
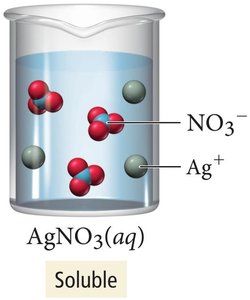
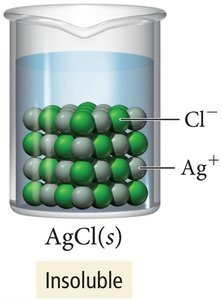
Soluble vs. Insoluble Salts
Some salts, like AgNO3, are highly soluble in water, while others, like AgCl, are insoluble and remain as solids.
Solubility Rules
Solubility rules are empirical guidelines developed from experimental data to predict whether an ionic compound will dissolve in water.
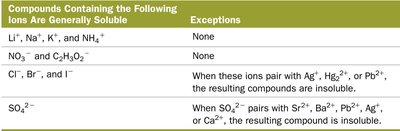
Compounds Containing the Following Ions Are Generally Soluble | Exceptions |
|---|---|
Li+, Na+, K+, NH4+ | None |
NO3-, C2H3O2- | None |
Cl-, Br-, I- | When paired with Ag+, Hg22+, or Pb2+, insoluble |
SO42- | When paired with Sr2+, Ba2+, Pb2+, Ag+, or Ca2+, insoluble |
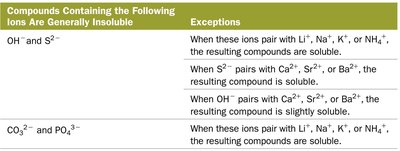
Compounds Containing the Following Ions Are Generally Insoluble | Exceptions |
|---|---|
OH-, S2- | When paired with Li+, Na+, K+, NH4+, soluble; S2- with Ca2+, Sr2+, Ba2+, soluble; OH- with Ca2+, Sr2+, Ba2+, slightly soluble |
CO32-, PO43- | When paired with Li+, Na+, K+, NH4+, soluble |
Summary
This guide covers the essential concepts of solutions, concentration, dilution, stoichiometry, solubility, and electrolytes in aqueous chemistry. Understanding these principles is crucial for predicting and analyzing chemical reactions in water.
