 Back
BackIntroduction to Solutions and Aqueous Reactions
Study Guide - Smart Notes

Solutions and Their Properties
Key Definitions and Concepts
Solutions are homogeneous mixtures composed of a solute dissolved in a solvent. Understanding the terminology and properties of solutions is essential for studying chemical reactions in aqueous environments.
Solute: The substance that is dissolved in a liquid.
Solvent: The liquid that dissolves the solute (often water in aqueous solutions).
Solution: The homogeneous mixture of solute and solvent.
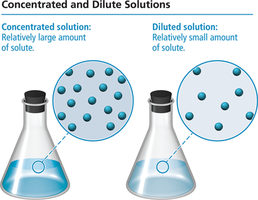
Concentrated solution: Contains a relatively large amount of solute.
Dilute solution: Contains a relatively small amount of solute.
Mixtures and Concentration
Mixtures can have variable composition. The concentration of a solution quantifies the amount of solute relative to the solvent. The most common unit of concentration in chemistry is molarity (M).
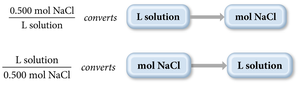
Molarity (M): The number of moles of solute per liter of solution.
Molarity Formula
Molarity is calculated using the following formula:
Preparing Solutions of Specified Concentration
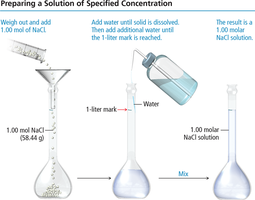
To prepare a solution of a specific molarity, follow these steps:
Weigh out the required amount of solute.
Add the solute to a volumetric flask.
Add water until the solute is dissolved, then fill to the calibration mark with water.
Mix thoroughly to ensure homogeneity.
Example: Preparing a Solution
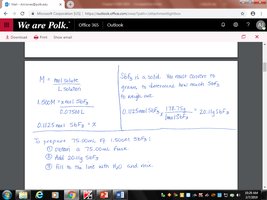
How would you prepare 75.00 mL of a 1.500 M solution of antimony fluoride (SbF3)?
Calculate the required moles of SbF3 using .
Convert moles to grams using the molar mass.
Dissolve the calculated mass in water and dilute to the final volume.
Solution Dilution
To prepare a less concentrated solution from a more concentrated stock solution, use the dilution equation:
and are the molarity and volume of the stock solution.
and are the molarity and volume of the diluted solution.
Example: To prepare 100.0 mL of 1.00 M HCl from 12.0 M HCl, calculate the required volume of stock solution and dilute with water to the final volume.
Types of Aqueous Solutions and Solubility
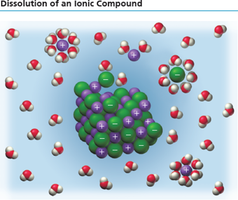
Ionic Theory of Solutions
When ionic compounds dissolve in water, they dissociate into ions, which are responsible for the solution's ability to conduct electricity. This concept was proposed by Arrhenius in 1884.
Example:
Deionized water does not conduct electricity because it lacks ions.
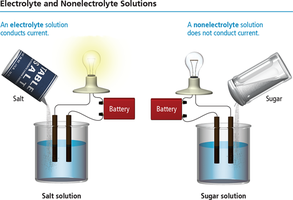
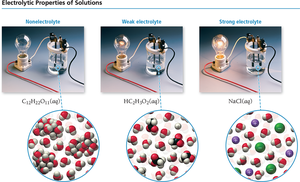
Electrolytes and Nonelectrolytes
Electrolytes are substances that produce ions in solution and conduct electricity. Nonelectrolytes do not produce ions and do not conduct electricity.
Strong electrolytes: Dissociate completely (e.g., soluble ionic compounds, strong acids, strong bases).
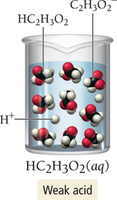
Weak electrolytes: Partially dissociate (e.g., weak acids and bases).
Nonelectrolytes: Do not dissociate (e.g., molecular compounds like sugar).
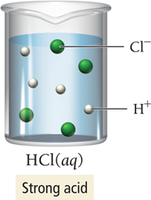
Strong vs. Weak Electrolytes
Strong electrolytes exist almost entirely as ions in solution, while weak electrolytes only partially ionize. Nonelectrolytes remain as molecules.
Example (strong):
Example (weak):
Solubility Rules and Precipitation Reactions
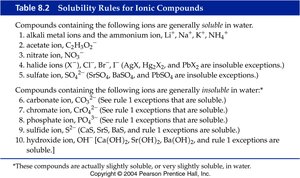
Solubility Rules
Solubility rules help predict whether an ionic compound will dissolve in water or form a precipitate. These rules are essential for predicting the outcomes of double displacement reactions.
Practice: Solubility Determination
Use solubility rules to determine if a compound is soluble or insoluble in water. For example, sodium bromide is soluble, while barium sulfate is insoluble.
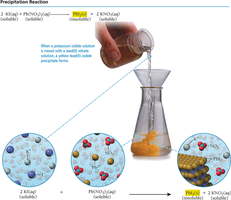
Precipitation Reactions
Precipitation reactions occur when two solutions are mixed and an insoluble solid (precipitate) forms. The solubility rules are used to predict the formation of a precipitate.
Write the possible products and their states.
If a product is insoluble, it will precipitate.
Balance the equation.
Writing Ionic Equations
There are three main types of equations for aqueous reactions:
Molecular equation: All substances written as compounds.
Complete ionic equation: Aqueous substances written as ions.
Net ionic equation: Only the species that change during the reaction are included; spectator ions are omitted.
Example: Reaction of magnesium nitrate and sodium chromate.
Acid-Base and Gas Evolution Reactions
Acid-Base (Neutralization) Reactions
Acid-base reactions involve the reaction of an acid and a base to produce water and an ionic salt. Acids produce hydronium ions (H+), while bases produce hydroxide ions (OH-).
General equation:
Gas Evolution Reactions
Gas evolution reactions produce a gas as one of the products, often observed as bubbling in the solution.
Types of Chemical Reactions
Classification of Reactions
Combination (Synthesis): Two or more reactants form one product.
Decomposition: One reactant breaks down into two or more products.
Single Replacement: One element replaces another in a compound.
Double Replacement: Cations and anions exchange partners.
Neutralization: Acid reacts with base to form water and a salt.
Activity Series and Single Replacement Reactions
The activity series ranks elements by their reactivity. A more active metal will replace a less active metal in a compound.
Check the activity series before predicting if a single replacement reaction will occur.
Properties of Acids and Bases
Acids
Produce hydrogen ions (H+).
Taste sour.
Turn blue litmus paper red.
Bases
Produce hydroxide ions (OH-).
Taste bitter; feel slippery.
Turn red litmus paper blue.
Brønsted-Lowry Theory
According to Brønsted-Lowry, an acid is a proton (H+) donor, and a base is a proton acceptor. Conjugate acid-base pairs differ by one proton.
Example:
Oxidation-Reduction (Redox) Reactions
Redox Concepts
Redox reactions involve the transfer of electrons between substances. Oxidation is the loss of electrons, and reduction is the gain of electrons.
Example:
Oxidation Numbers
Oxidation numbers indicate the charge an atom would have if electrons were assigned according to certain rules:
Free elements have an oxidation number of 0.
Monoatomic ions have an oxidation number equal to their charge.
Hydrogen is usually +1; oxygen is usually -2.
The sum of oxidation numbers in a compound is 0; in a polyatomic ion, it equals the ion's charge.
Half-Reaction Method
Redox reactions can be split into two half-reactions: one for oxidation and one for reduction. Each half-reaction is balanced for mass and charge, then combined to give the overall balanced equation.
Balance all atoms except O and H.
Balance O using H2O, H using H+.
Balance charge by adding electrons.
Multiply half-reactions to equalize electrons, then add and cancel identical species.
