 Back
BackLewis Structures and Molecular Geometry: A Comprehensive Study Guide
Study Guide - Smart Notes

Lewis Structures and Molecular Geometry
Introduction to Lewis Structures
Lewis structures are diagrams that represent the bonding between atoms of a molecule and the lone pairs of electrons that may exist. They are foundational for understanding molecular structure, bonding, and reactivity in general chemistry.
Valence electrons are the outermost electrons involved in bonding.
Lewis structures help visualize how atoms share or transfer electrons to achieve stable electron configurations, often resembling those of noble gases.

Electron Configuration and Valence Electrons
Electron configurations describe the arrangement of electrons in an atom. Noble gas notation simplifies this by using the symbol of the nearest noble gas to represent core electrons.
Example: Sodium (Na, Z=11): [Ne]3s1
Valence electrons are those in the outermost shell (s and p orbitals for main group elements).
General Guidelines for Drawing Lewis Structures
To draw accurate Lewis structures, follow these steps:
Count all valence electrons for the molecule or ion.
Arrange atoms with the least electronegative atom (except H) in the center.
Connect atoms with single bonds first, then add lone pairs to complete octets (or duets for H).
Use double or triple bonds if necessary to satisfy the octet rule.
Assign formal charges to minimize nonzero values, favoring negative charges on electronegative atoms.
Formal Charge Formula:
Main Group Trends in Lewis Structures
The number of bonds and lone pairs for main group elements follows periodic trends:
Group | 1A | 2A | 3A | 4A | 5A | 6A | 7A | 8A |
|---|---|---|---|---|---|---|---|---|
# Bonds | 1 | 2 | 3 | 4 | 3 | 2 | 1 | 0 |
# Lone Pairs | 0 | 0 | 0 | 0 | 1 | 2 | 3 | 4 |
Single, Double, and Triple Bonds
Atoms can share one, two, or three pairs of electrons, forming single, double, or triple bonds, respectively. The number of shared pairs determines the bond order.
Single bond: 1 shared pair (e.g., F2)
Double bond: 2 shared pairs (e.g., O2)
Triple bond: 3 shared pairs (e.g., N2)
Lewis Structures of Simple Molecules
Examples of common molecules with their Lewis structures:
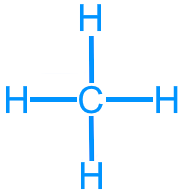
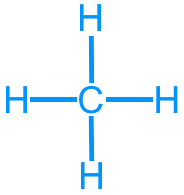
Methane (CH4): 8 valence electrons
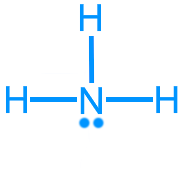
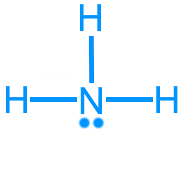
Ammonia (NH3): 8 valence electrons
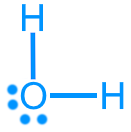
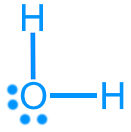
Water (H2O): 8 valence electrons
Lewis Structures of Multi-Central Atom Molecules
Some molecules have more than one central atom, requiring careful placement of atoms and electrons.
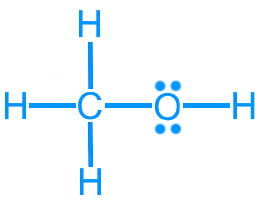
Methanol (CH3OH): 14 valence electrons
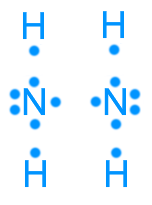
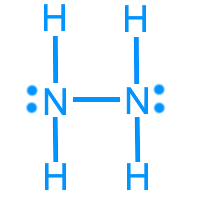
Hydrazine (N2H4): 14 valence electrons
Lewis Structures of Row 3 Central Atoms and Hypervalency
Elements in period 3 and beyond can have expanded octets (hypervalency), accommodating more than 8 electrons.
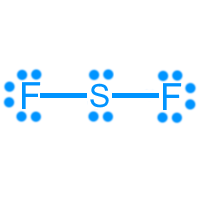
Sulfur difluoride (SF2): 20 valence electrons
Sulfur tetrafluoride (SF4): 34 valence electrons (hypervalent)
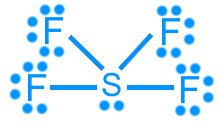
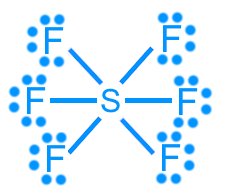
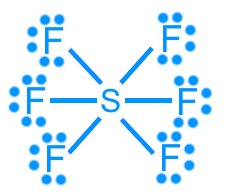
Sulfur hexafluoride (SF6): 48 valence electrons (hypervalent)
Lewis Structures of Polyatomic Ions and Formal Charge
For ions, add electrons for negative charges and subtract for positive charges. Formal charges help identify the most stable structure.
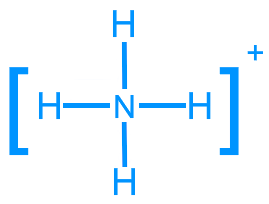
Ammonium (NH4+): 8 valence electrons
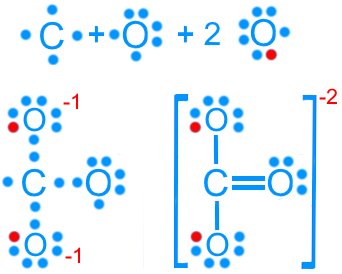
Carbonate (CO32–): 24 valence electrons
Resonance Structures
Some molecules cannot be represented by a single Lewis structure. Resonance structures are multiple valid Lewis structures that differ only in the placement of electrons, not atoms.
Carbonate (CO32–): Three resonance forms
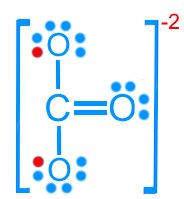
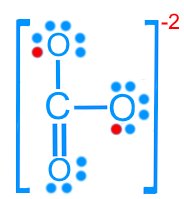
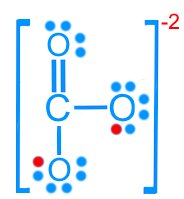
Resonance stabilizes molecules by delocalizing electrons, resulting in fractional bond orders and charges.
Bond Strength, Polarity, and Molecular Geometry
Bond Order, Length, and Strength
Bond order is the number of shared electron pairs between two atoms. It affects bond length and strength:
As bond order increases, bond length decreases.
As bond order increases, bond strength increases.
Examples:
Bond | Bond Order |
|---|---|
C–C | 1 |
C=C | 2 |
C≡C | 3 |
Bond Polarity and Electronegativity
Bond polarity arises from differences in electronegativity between bonded atoms. The more electronegative atom attracts electrons more strongly, resulting in partial charges (δ+ and δ–).
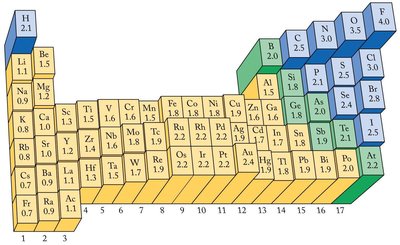
Nonpolar bonds: Equal sharing of electrons (e.g., N–N).
Polar bonds: Unequal sharing, leading to dipoles (e.g., H–F).
VSEPR Theory and AXE Notation
Valence Shell Electron Pair Repulsion (VSEPR) theory predicts molecular shapes based on electron pair repulsion. AXE notation summarizes the number of atoms (X) and lone pairs (E) around a central atom (A).
A: Central atom
X: Number of bonded atoms
E: Number of lone pairs on the central atom
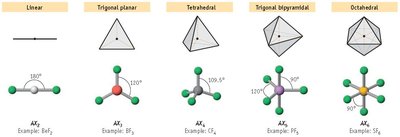
Examples of Electron Pair and Molecular Geometry
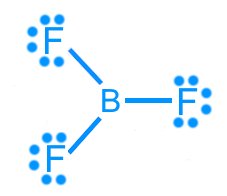
Boron trifluoride (BF3): AX3, trigonal planar, 120°, nonpolar
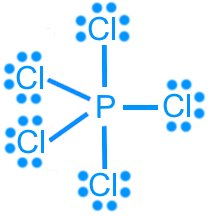
Phosphorus pentachloride (PCl5): AX5, trigonal bipyramidal, 120°/90°, nonpolar
Sulfur hexafluoride (SF6): AX6, octahedral, 90°, nonpolar
Electron Pair vs. Molecular Geometry: Common Examples
Methane (CH4): AX4, tetrahedral, 109.5°, nonpolar
Ammonia (NH3): AX3E, trigonal pyramidal, ~107°, polar
Water (H2O): AX2E2, bent, ~104.5°, polar
Applying VSEPR to Larger Molecules
For larger molecules, analyze each central atom separately using AXE notation and consider the overall molecular polarity.
Propane (C3H8): All central carbons are AX4 (tetrahedral), nonpolar overall.
Acetone (C3H6O): Oxygen is AXE2 (linear), carbonyl carbon is AX3 (trigonal planar); molecule is polar due to C=O bond.
Aspirin (C9H8O4): Contains both polar and nonpolar regions; benzene ring is nonpolar, carboxyl and ester groups are polar.
Additional info: For each molecule, lone pairs on heteroatoms (O, N) must be included to accurately predict geometry and polarity. Resonance and formal charge analysis are essential for ions and delocalized systems.
