 Back
BackLiquids and Solids: Intermolecular Forces, Physical Properties, and Phase Changes
Study Guide - Smart Notes

Liquids and Solids
Introduction to Intermolecular Forces
Liquids and solids are condensed phases of matter, and their properties are largely determined by the forces acting between molecules, known as intermolecular forces. These forces are distinct from the stronger intramolecular forces (chemical bonds) that hold atoms together within a molecule.
Intramolecular forces: Bonds within a molecule (e.g., covalent bonds).
Intermolecular forces: Attractions between molecules.
Types of Intermolecular Forces
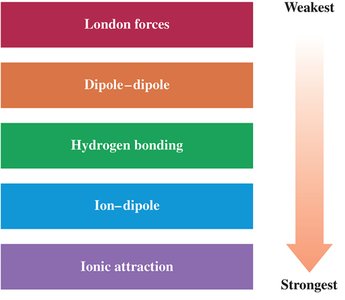
There are several types of intermolecular forces, each with different strengths and characteristics. The main types, in order of increasing strength, are:
Type | Description |
|---|---|
London Dispersion Forces | Weak, temporary attractions due to momentary dipoles in all atoms and molecules. |
Dipole–Dipole Forces | Attractions between permanent dipoles in polar molecules. |
Hydrogen Bonding | Strong dipole–dipole interaction when H is bonded to N, O, or F. |
Ion–Dipole Forces | Attraction between an ion and a polar molecule. |
Ionic Attraction | Strongest; between full charges of cations and anions. |
London Dispersion Forces
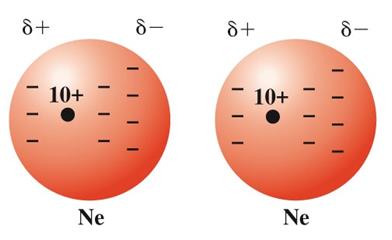
London dispersion forces arise from temporary fluctuations in electron distribution, creating instantaneous dipoles that induce dipoles in neighboring atoms or molecules. These forces are present in all substances but are the only intermolecular force in nonpolar molecules.
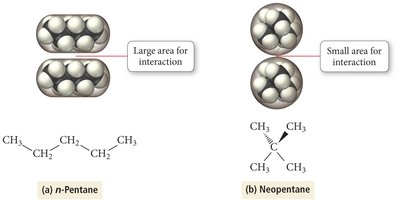
Strength increases with molecular size and surface area.
Linear molecules have stronger dispersion forces than branched ones.
Name | Structure | Boiling Point (°C) |
|---|---|---|
Methane | CH4 | -161 |
Ethane | CH3CH3 | -89 |
Propane | -42 | |
Butane | -0.5 | |
Pentane | 36 | |
Hexane | 69 | |
Heptane | 98 | |
Octane | 125 | |
Nonane | 151 | |
Decane | 174 |
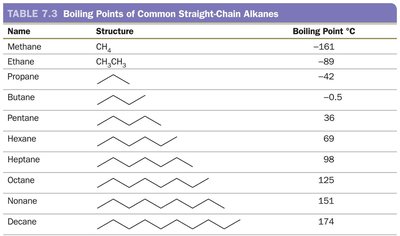
Dipole–Dipole Forces
Dipole–dipole forces occur between polar molecules, where the positive end of one molecule is attracted to the negative end of another. These forces are stronger than London dispersion forces due to the presence of permanent dipoles.

Only present in polar molecules.
Responsible for higher boiling points compared to nonpolar molecules of similar size.
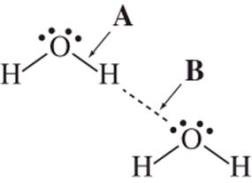
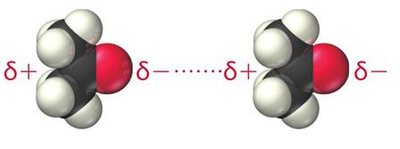
Hydrogen Bonding
Hydrogen bonding is a special, strong type of dipole–dipole interaction that occurs when hydrogen is bonded to highly electronegative atoms (N, O, or F). The hydrogen atom acts as a bridge between two electronegative atoms.
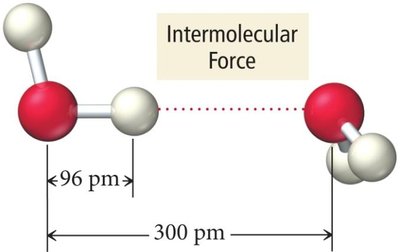
Name | Description |
|---|---|
Hydrogen-bond donor (δ+) | A molecule with a hydrogen atom covalently bonded to O, N, or F. |
Hydrogen-bond acceptor (δ−) | A molecule with a lone pair on O, N, or F. |

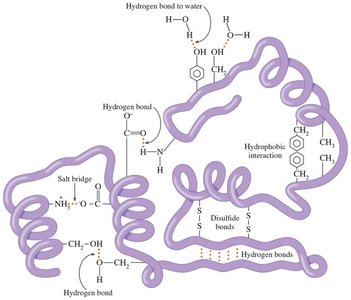
Hydrogen bonds are crucial in biological molecules, such as DNA base pairing.
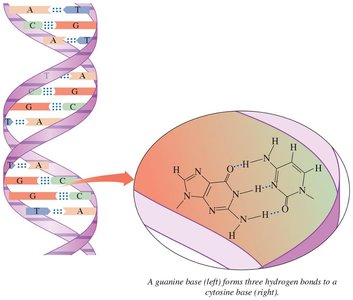
Hydrogen bonding explains the anomalously high boiling point of water compared to other group 16 hydrides.
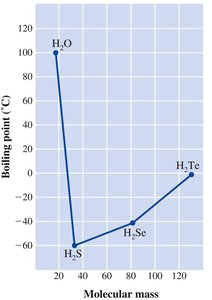
Ion–Dipole and Ionic Attractions
Ion–dipole forces occur when an ion interacts with a polar molecule, important in solutions of ionic compounds in water. Ionic attractions (ionic bonds) are the strongest, occurring between cations and anions in ionic solids.

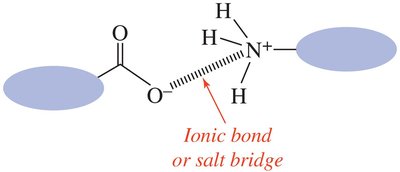
Ionic attractions are responsible for the structure of ionic solids and salt bridges in proteins.
Molecular Polarity and Determining Intermolecular Forces
The polarity of a molecule depends on both the electronegativity of the atoms and the molecular geometry. For molecules with more than two atoms, consider both bond polarity and shape.
If the central atom has lone pairs, the molecule is usually polar.
If all atoms bonded to the central atom are the same and there are no lone pairs, the molecule is nonpolar.
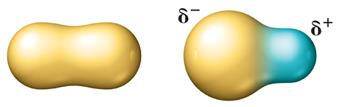
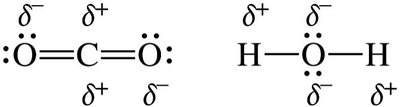
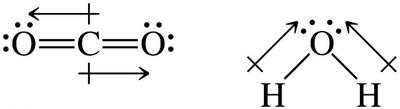
To determine the strongest intermolecular force present:
Is the molecule polar? If no, London forces dominate. If yes, continue.
Does the molecule contain N–H, O–H, or F–H? If yes, hydrogen bonding is present. If no, dipole–dipole forces are strongest.
If ions are present, consider ion–dipole or ionic forces.
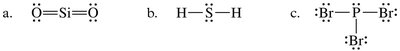
Consequences of Intermolecular Forces: Physical Properties
The strength and type of intermolecular forces affect many physical properties of substances:
Boiling and melting points: Stronger forces lead to higher boiling/melting points.
Solubility: "Like dissolves like"—polar substances dissolve in polar solvents, nonpolar in nonpolar.
Surface tension: Energy required to increase the surface area of a liquid; higher with stronger forces.
Viscosity: Resistance to flow; higher with stronger forces.
Vapor pressure: Pressure of vapor above a liquid at equilibrium; lower with stronger forces.

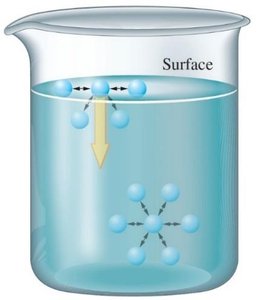



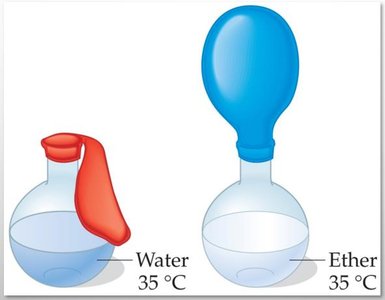
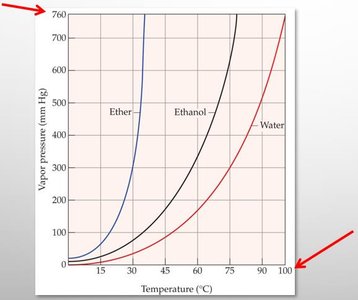
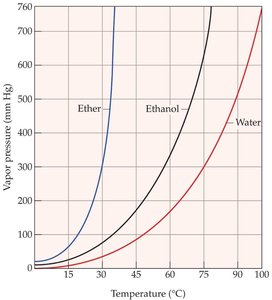
Vapor Pressure and Boiling Point
Liquids evaporate as molecules escape from the surface. The vapor pressure is the equilibrium pressure of vapor above the liquid at a given temperature. Stronger intermolecular forces result in lower vapor pressure and higher boiling points.
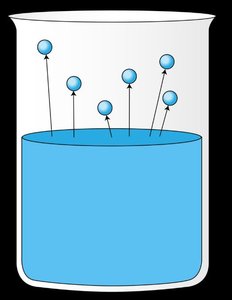
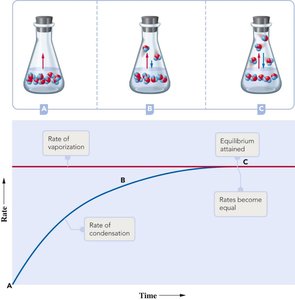
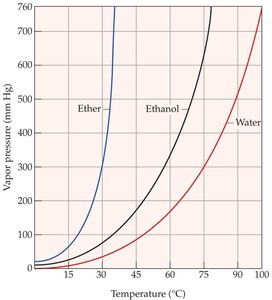
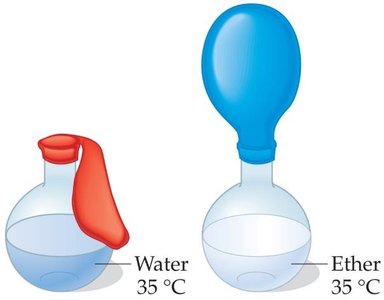
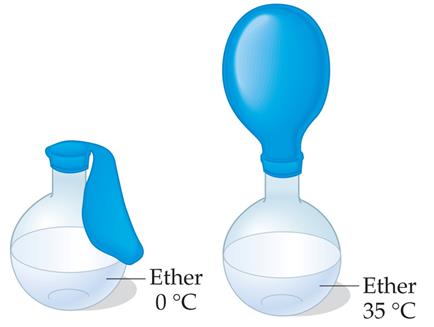
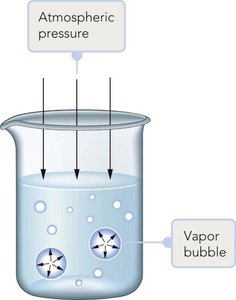
The boiling point is the temperature at which the vapor pressure equals the external pressure. Lower external pressure leads to a lower boiling point.
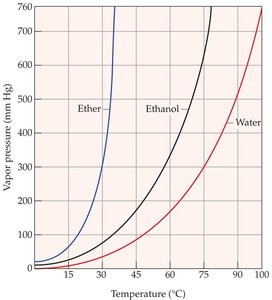
Clausius–Clapeyron Equation
The Clausius–Clapeyron equation quantitatively relates vapor pressure and temperature:
For two temperatures and pressures:
= enthalpy of vaporization (J/mol)
= gas constant (8.314 J/(mol·K))
= temperature in Kelvin
Types of Solids
Solids can be classified as crystalline (ordered, repeating structure) or amorphous (disordered, random arrangement). Crystalline solids are further classified by their unit cell structure.
Cubic Unit Cells
Cubic unit cells are the simplest and most common. Atoms can be located at corners, faces, edges, or the center of the cell. The number of atoms per unit cell depends on their positions.
Corner atom: shared by 8 cells (1/8 per cell)
Edge atom: shared by 4 cells (1/4 per cell)
Face atom: shared by 2 cells (1/2 per cell)
Empirical Formula from Unit Cell
To determine the empirical formula, count the total number of each type of atom in the unit cell, considering sharing between cells.
Position | Number per cell | Fraction in cell | Total per cell |
|---|---|---|---|
Corner | 8 | 1/8 | 1 |
Face | 6 | 1/2 | 3 |
Edge | 12 | 1/4 | 3 |
Center | 1 | 1 | 1 |
Types of Cubic Cells
Simple cubic: 1 atom per cell
Body-centered cubic: 2 atoms per cell
Face-centered cubic: 4 atoms per cell
Atomic Radius Calculation in FCC Unit Cells
For a face-centered cubic (FCC) unit cell, the edge length is related to the atomic radius by .
Example: Gold (Au) crystallizes in an FCC unit cell with a density of 19.3 g/cm3 and atomic mass 197.0 g/mol. Calculate the atomic radius in pm.
Steps:
Calculate the volume of the unit cell using density and mass.
Find edge length from the volume.
Relate to using .
Phase Changes and Phase Diagrams
A phase is a homogeneous part of a system separated by boundaries. Phase changes are physical changes between solid, liquid, and gas. A phase diagram shows the conditions under which each phase exists.
Triple point: All three phases coexist in equilibrium.
Critical point: Above this temperature, the liquid phase does not exist.
Heating and Cooling Curves
Heating curves show temperature changes as a substance is heated. During phase changes, temperature remains constant as energy is used for the phase transition.
For temperature changes:
For phase changes:
Example: Calculate the heat required to convert 135.0 g of ice at –15.0°C to water vapor at 120.0°C, using the appropriate and values for each phase and transition.
Summary Table: Physical Properties and Intermolecular Forces
Property | Effect of Stronger Intermolecular Forces |
|---|---|
Surface tension | Increases |
Viscosity | Increases |
Vapor pressure | Decreases |
Boiling point | Increases |
Melting point | Increases |
Practice Problems
Rank molecules by vapor pressure and boiling point based on intermolecular forces.
Interpret phase diagrams to determine normal boiling/melting points, triple point, and physical state at given conditions.
Calculate heat required for multi-step phase changes using and .
Additional info: For further practice, refer to end-of-chapter problems and online resources as recommended in your course materials.
