 Back
BackLiquids and Solids: Intermolecular Forces, Properties, and Structures
Study Guide - Smart Notes

Liquids and Solids: Intermolecular Forces, Properties, and Structures
Intermolecular vs. Intramolecular Forces
Understanding the distinction between intramolecular and intermolecular forces is fundamental to predicting the behavior of substances in the liquid and solid states.
Intramolecular forces are the covalent bonds that hold atoms together within a molecule.
Intermolecular forces (IMF) are weaker, temporary attractions between different molecules or particles in a sample.
IMFs are responsible for many physical properties of substances, such as melting and boiling points, viscosity, surface tension, and solubility.
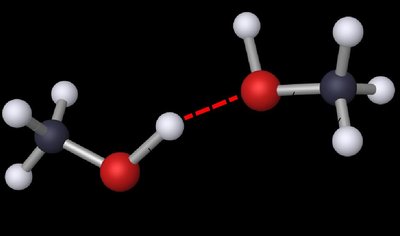
Types of Intermolecular Forces
There are several types of intermolecular forces, each with distinct characteristics and effects on physical properties.
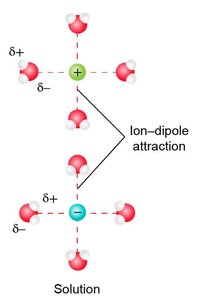
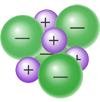
Ion–dipole attractions: Occur between ions and polar molecules; strongest IMF.
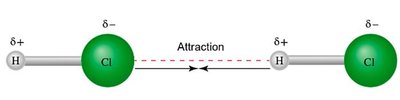
Dipole–dipole attractions: Occur between polar molecules.
Hydrogen bonding: A special, strong type of dipole–dipole attraction involving H bonded to N, O, or F.
Dispersion forces (London forces): Present in all molecules, especially nonpolar ones; arise from temporary dipoles.
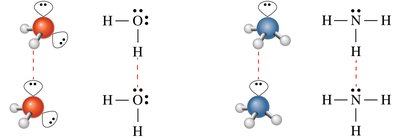
Hydrogen Bonding
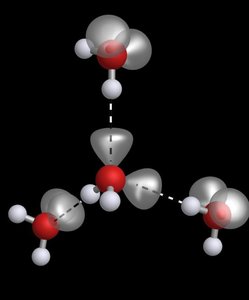
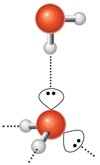
Hydrogen bonding is a particularly important intermolecular force, responsible for many unique properties of water and biological molecules.
Requires a hydrogen bond donor (H bonded to N, O, or F) and an acceptor (N, O, or F with lone pairs).
Hydrogen bonds are much stronger than typical dipole–dipole interactions.
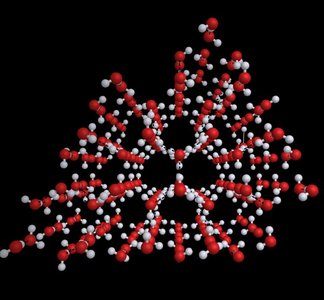
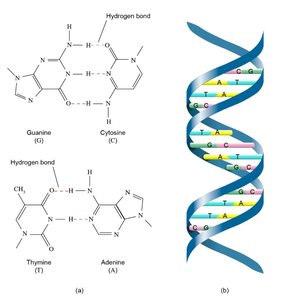
Responsible for the low density of ice, the structure of DNA, and the shapes of proteins.

Hydrogen Bonding in Water and Biological Molecules
Hydrogen bonding leads to anomalous properties in water and is crucial in biological systems.
Ice is less dense than liquid water due to hydrogen bonding, allowing it to float.
Hydrogen bonds stabilize the double helix structure of DNA and the folding of proteins.
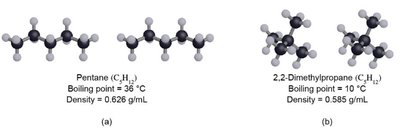
Dispersion Forces (London Forces)
Dispersion forces are present in all molecules and arise from instantaneous dipoles caused by random electron movement.
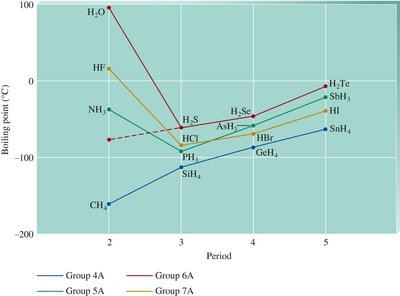
Strength increases with atomic size, number of electrons, and molecular complexity.
Responsible for the boiling points of nonpolar substances.
Ranking Intermolecular Forces
The strength of intermolecular forces determines many physical properties.
Intermolecular Force | Present In | Example(s) |
|---|---|---|
Ion–dipole | Mixtures of ionic and polar covalent compounds | Salt water (NaCl in H2O) |
Hydrogen bonding | Substances with H bonded to N, O, or F | H2O |
Dipole–dipole | Polar molecules | H2O, HCl |
Dispersion | All molecular substances | H2O, HCl, Cl2 |
Properties of Liquids
The physical properties of liquids are determined by the strength and type of intermolecular forces present.
Viscosity: Resistance to flow; decreases with increasing temperature.
Surface tension: Tendency of a liquid to minimize its surface area; caused by cohesive forces among molecules.
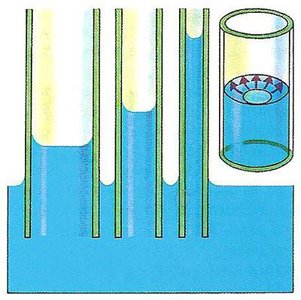
Capillary action: Ability of a liquid to flow against gravity in narrow tubes due to adhesion and cohesion.
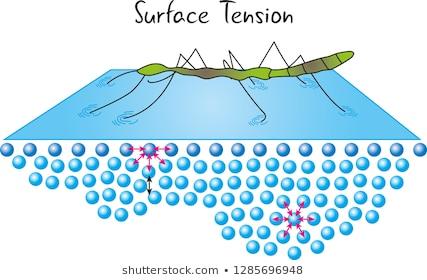

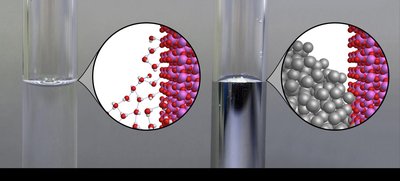
Cohesion, Adhesion, and Capillary Action
Cohesion and adhesion are important for understanding liquid behavior in various contexts.
Cohesion: Attraction between like particles (e.g., water molecules).
Adhesion: Attraction between different particles (e.g., water and glass).
Capillary action: Results from both cohesion and adhesion, allowing liquids to rise in narrow tubes.


Phase Changes and Phase Diagrams
Phase changes involve transitions between solid, liquid, and gas states, each requiring or releasing energy.
Melting, freezing, vaporization, condensation, sublimation, deposition are key phase transitions.
Phase diagrams show the state of a substance at various temperatures and pressures.
The triple point is where all three phases coexist; the critical point marks the end of the liquid-gas boundary.
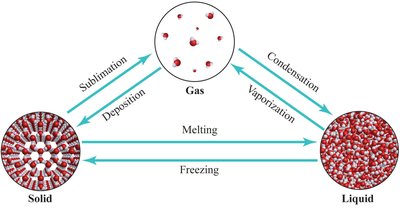
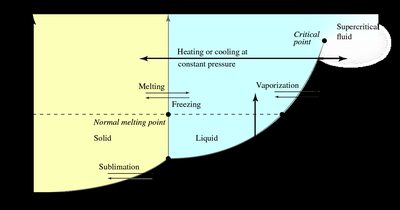
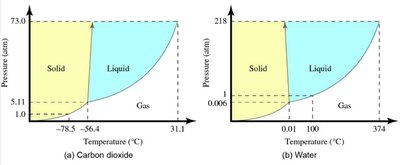
Heating Curves and Enthalpy of Phase Changes
Heating curves illustrate how temperature changes as a substance is heated, showing plateaus during phase changes.
Energy required for heating (no phase change):
Energy required for phase change: or
Heats of fusion (), vaporization (), and sublimation () are key values.
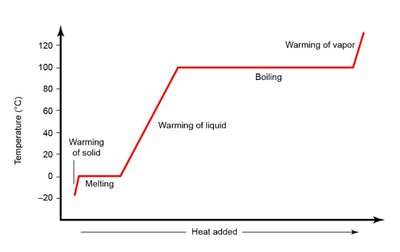
Vapor Pressure, Boiling Point, and Clausius–Clapeyron Equation
Vapor pressure is the pressure exerted by a vapor in equilibrium with its liquid. The boiling point is when vapor pressure equals atmospheric pressure.
Volatile substances have high vapor pressures; nonvolatile substances have low vapor pressures.
Clausius–Clapeyron equation relates vapor pressure and temperature:
Two-point version:
Boiling and Distillation
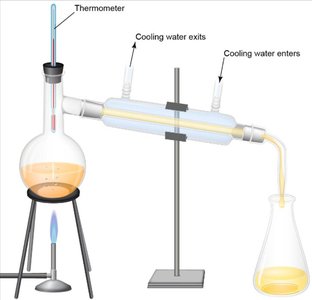
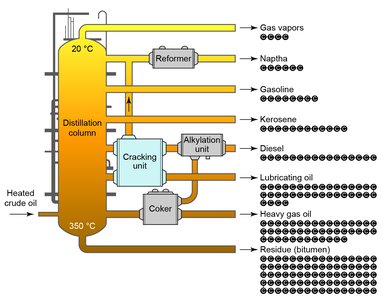
Boiling occurs when the vapor pressure of a liquid equals the external pressure. Distillation is used to separate volatile substances based on their boiling points.
Normal boiling point: boiling at 1 atm pressure.
Distillation apparatus separates components by vaporizing and condensing them.

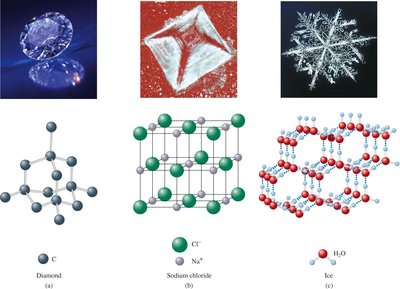
Solids: Classification and Structure
Solids are classified based on the organization of their particles and the nature of their bonding.
Crystalline solids: Have a regular, repeating structure and definite melting points (e.g., ice, NaCl).
Amorphous solids: Lack a regular structure and soften gradually (e.g., glass, chocolate).
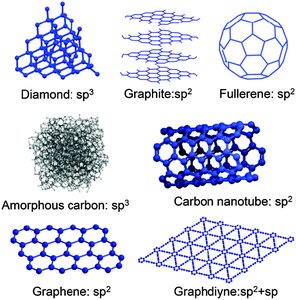
Allotropes and Metallic Bonding
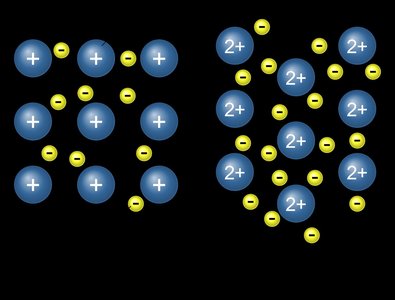
Allotropes are different structural forms of the same element. Metallic solids are characterized by a sea of delocalized electrons.
Metals are malleable, ductile, and conduct electricity due to mobile electrons.
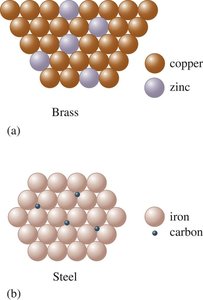
Alloys are mixtures of metals; can be substitutional (brass) or interstitial (steel).
Unit Cells and Crystal Structures
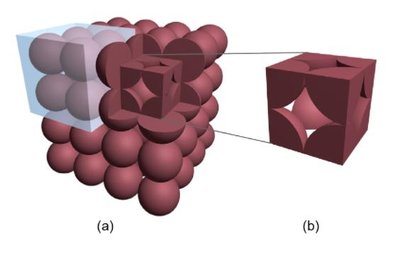
The unit cell is the simplest repeating unit in a crystal. Cubic unit cells are common and include simple cubic, body-centered cubic (bcc), and face-centered cubic (fcc).
Simple cubic: 1 atom/unit cell, 52% packing efficiency, coordination number 6,
Body-centered cubic: 2 atoms/unit cell, 68% packing efficiency, coordination number 8,
Face-centered cubic: 4 atoms/unit cell, 74% packing efficiency, coordination number 12,
Unit Cell | Atoms/Unit Cell | Packing Efficiency | Coordination Number | Edge Length and Radius |
|---|---|---|---|---|
Simple cubic | 1 | 52% | 6 | |
Body-centered cubic | 2 | 68% | 8 | |
Face-centered cubic | 4 | 74% | 12 |
Special Solids: Nitinol and Packing Variations
Nitinol is an alloy with memory-shape properties due to reversible solid-to-solid phase changes. Different packing arrangements lead to various crystal structures.
Additional info: These notes cover the essential concepts from Chapter 10: Liquids and Solids, including intermolecular forces, properties of liquids, phase changes, and the structure of solids. All images included are directly relevant to the explanations provided.
