 Back
BackLiquids and Solids: Intermolecular Forces, Properties, and Phase Changes
Study Guide - Smart Notes

Liquids and Solids
Intermolecular Forces (IMF)
Intermolecular forces are the attractions between molecules that determine many physical properties of substances, such as boiling point, melting point, surface tension, and viscosity. These forces are generally much weaker than intramolecular (chemical) bonds, but they play a crucial role in the behavior of liquids and solids.
Intramolecular forces: Forces within a molecule, such as covalent and ionic bonds.
Intermolecular forces: Forces between molecules, including dipole-dipole, hydrogen bonding, and London dispersion forces.
Measurement of IMF: Properties like boiling point, melting point, and heats of vaporization/fusion/sublimation reflect IMF strength.
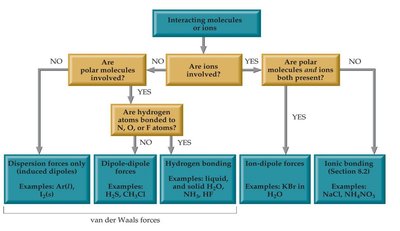
Types of Intermolecular Forces
Dipole–Dipole Forces: Occur between polar molecules with permanent dipoles. The positive end of one molecule is attracted to the negative end of another. The strength decreases with increasing distance between dipoles.
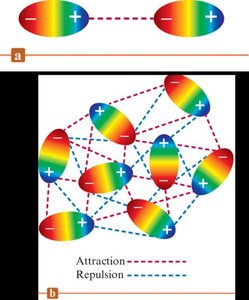
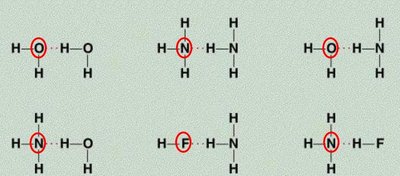
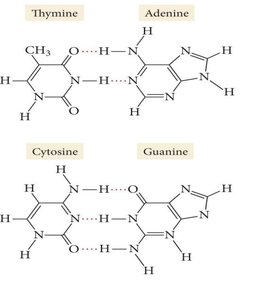
Hydrogen Bonding: A special, strong type of dipole-dipole force. Occurs when hydrogen is bonded to highly electronegative atoms (N, O, F) and interacts with another N, O, or F atom. Hydrogen bonding is responsible for many unique properties of water and biological molecules like DNA.
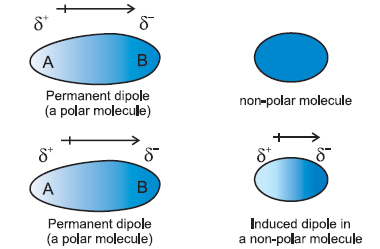
London Dispersion Forces: Present in all molecules and atoms, these arise from temporary dipoles due to momentary uneven electron distribution. They are the only IMF in nonpolar molecules and noble gases. The strength increases with molar mass and surface area.
Ion–Dipole Forces: Occur in mixtures of ionic compounds and polar molecules. These are the strongest intermolecular attractions and are especially important in aqueous solutions.
Summary Table: Types of Intermolecular Forces
Type | Occurs Between | Relative Strength | Example |
|---|---|---|---|
Dispersion (London) | All molecules/atoms | Weakest | Ar, F2 |
Dipole–Dipole | Polar molecules | Moderate | HCl, CH2Cl2 |
Hydrogen Bonding | H bonded to N, O, F | Strong | H2O, NH3 |
Ion–Dipole | Ions and polar molecules | Strongest | Na+ in H2O |
Polarity and Molecular Geometry
The polarity of a molecule depends on both the polarity of its bonds and its molecular geometry. Polar molecules have separated centers of positive and negative charge, while nonpolar molecules do not.
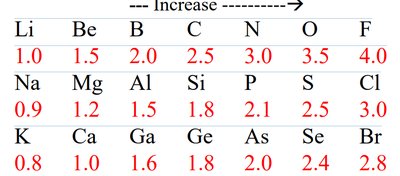
Electronegativity: The ability of an atom to attract electrons in a bond. Differences in electronegativity determine bond polarity.
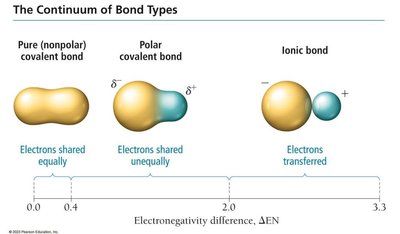
Bond Types: Pure covalent (nonpolar), polar covalent, and ionic bonds exist on a continuum based on electronegativity difference.
Dipole Moment: A quantitative measure of molecular polarity, defined as (charge times distance).
Physical Properties of Liquids
IMFs affect several key properties of liquids:
Surface Tension: Resistance to increasing surface area. Liquids with strong IMFs have high surface tension, forming spherical drops.

Capillary Action: The ability of a liquid to rise in a narrow tube, due to cohesive and adhesive forces. Polar liquids like water exhibit strong capillary action.
Meniscus: The shape of the liquid surface in a tube depends on the balance of adhesive and cohesive forces. Water forms a concave meniscus; mercury forms a convex meniscus.

Viscosity: Resistance to flow. Liquids with strong IMFs and complex molecules are more viscous. Increasing temperature decreases viscosity.
Boiling and Melting Points: Stronger IMFs result in higher boiling and melting points, as more energy is required to separate molecules.
Phase Changes and Energetics
Phase changes involve energy transfer without breaking chemical bonds. The temperature remains constant during a phase change.
Heat of Vaporization (): Energy required to vaporize one mole of liquid.
Heat of Fusion (): Energy required to melt one mole of solid.
Heat of Sublimation (): Energy required to convert one mole of solid directly to gas.
Heating/Cooling Curves: Show temperature changes and phase transitions as heat is added or removed.
Vaporization, Condensation, and Vapor Pressure
Vaporization is the process by which molecules escape from the liquid phase to the gas phase. Only molecules with sufficient kinetic energy can escape. As temperature increases, more molecules have enough energy to vaporize.
Vapor Pressure: The pressure exerted by vapor in equilibrium with its liquid. Higher temperature and weaker IMFs result in higher vapor pressure.
Boiling Point: The temperature at which vapor pressure equals external pressure. Lower external pressure lowers boiling point.
Clausius–Clapeyron Equation: Relates vapor pressure and temperature:
Classification of Solids
Solids are classified based on the nature of their constituent particles and the forces holding them together.
Type | Particles | Forces | Physical Properties |
|---|---|---|---|
Molecular | Molecules | London, dipole-dipole, H-bonding | Low mp, may sublime, poor heat/electric cond. |
Ionic | Ions | Ionic bonds | High mp, hard, brittle, solid noncond., liquid cond. |
Metallic | Atoms | Delocalized covalent bonds | Variable mp/hardness, good cond., malleable |
Network | Atoms | Covalent bonds | Very high mp, hard, brittle, non/semi cond. |
Phase Diagrams
Phase diagrams show the state of a substance at various temperatures and pressures. Key features include:
Regions: Represent solid, liquid, and gas states.
Lines: Indicate conditions where two phases coexist in equilibrium.
Triple Point: All three phases coexist.
Critical Point: The temperature and pressure above which a gas cannot be liquefied.
Supercritical Fluid: Properties of both gas and liquid above the critical point.
Summary of Key Concepts
IMFs determine the physical properties and phase behavior of substances.
Hydrogen bonding is especially important in water and biological molecules.
Phase changes require energy but do not break chemical bonds.
Vapor pressure and boiling point are influenced by temperature and IMF strength.
Solids are classified by their constituent particles and bonding types.
Phase diagrams summarize the conditions for different states and transitions.
