 Back
BackLiquids, Solids, and Intermolecular Forces: Chapter 12 Study Guide
Study Guide - Smart Notes

Liquids, Solids, and Gases
States of Matter and Their Properties
The three primary states of matter—solid, liquid, and gas—differ in particle arrangement, compressibility, and ability to flow. These differences are governed by the strength of intermolecular forces present in each state.
Solids: Particles are tightly packed and fixed in place. Solids are incompressible, retain their shape, and do not flow.
Liquids: Particles are close together but mobile. Liquids flow, take the shape of their container, but do not expand or compress easily.
Gases: Particles are far apart and move freely. Gases compress, expand, and flow easily.
Intermolecular Forces: The strength of these forces determines the physical properties and behavior of each state.


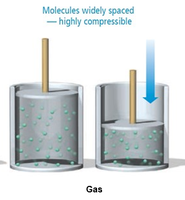
Intermolecular Forces
Types and Effects of Intermolecular Forces
Intermolecular forces are the attractions between particles (atoms, molecules, ions) that determine a substance’s physical properties such as boiling point, melting point, vapor pressure, and physical state. There are four main types:
Dispersion Force (London Force): Weakest force, present in all molecules and atoms, especially significant in non-polar substances.
Dipole–Dipole Force: Occurs between polar molecules due to permanent dipoles.
Hydrogen Bonding: Strongest in pure molecular substances, occurs when hydrogen is bonded to F, O, or N.
Ion–Dipole Force: Strongest overall, occurs in mixtures of ionic compounds and polar molecules.
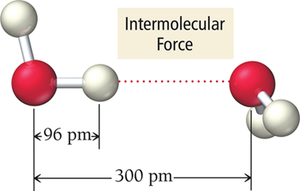
Dispersion Forces
Dispersion forces arise from temporary dipoles created by fluctuations in electron distribution. These forces are present in all atoms and molecules but are most significant in non-polar substances.
Temporary Dipole: Regions of excess electron density become partially negative (δ−), while depleted regions become partially positive (δ+).
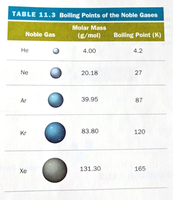
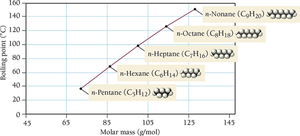
Strength Factors: Larger molar mass and more surface-to-surface contact increase dispersion force strength.
Examples: He, Cl2, CH4, C6H6.
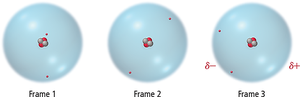
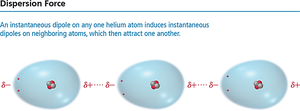
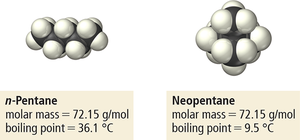
Effect of Molecular Size and Shape on Dispersion Forces
The strength of dispersion forces increases with molecular size (molar mass) and shape.
Size: Larger molecules have more electrons, leading to greater polarizability and stronger attractions.
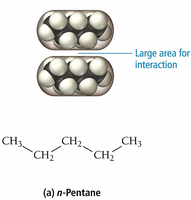
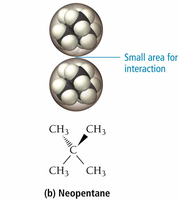
Shape: Straight-chain molecules have more surface contact and stronger forces than branched molecules.
Dipole–Dipole Forces
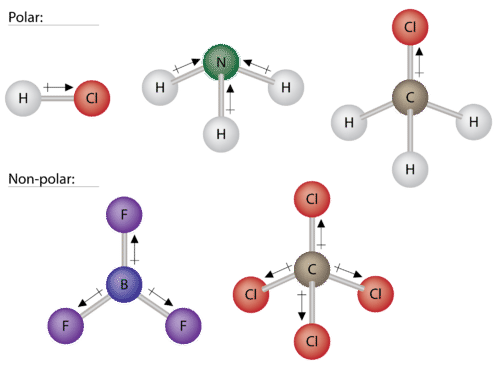
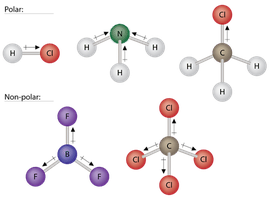
Dipole–dipole forces occur between polar molecules due to permanent dipoles arising from differences in electronegativity. The more electronegative atom attracts electrons, creating partial charges that lead to intermolecular attraction.
Polarity: Molecules like HCl, NH3, and CH3Cl are polar; BF3 and CCl4 are nonpolar despite polar bonds.
Strength: Dipole–dipole forces are stronger than dispersion forces and depend on electronegativity and molecular structure.
Example: Formaldehyde (CH2O) has a higher boiling point than ethane (C2H6).
Hydrogen Bonding
Hydrogen Bonding in Molecular Substances
Hydrogen bonding occurs among polar molecules where hydrogen is bonded to fluorine, oxygen, or nitrogen. The strong partial positive charge on hydrogen attracts lone pairs on nearby molecules, making hydrogen bonds stronger than dipole–dipole or dispersion forces.
Examples: HF, H2O, NH3.
Effect: Raises boiling points significantly (e.g., water boils at 100°C).
Ion–Dipole Forces
Ion–Dipole Forces in Mixtures
Ion–dipole forces occur when an ionic compound is mixed with a polar molecule. Positive ions attract negative dipoles, and negative ions attract positive dipoles. This force is key in dissolving ionic compounds in water and is the strongest intermolecular force.
Example: NaCl dissolving in water.
Intermolecular Forces in Action
Surface Tension
Surface tension is the tendency of a liquid to minimize its surface area due to cohesive forces. Molecules at the surface experience a net inward pull, creating a skin-like layer. Surface tension increases with stronger intermolecular forces and decreases with temperature.
Example: Water has higher surface tension than benzene.
Viscosity
Viscosity is the resistance of a liquid to flow, measured in centipoise (cP). It increases with intermolecular force and molar mass, and decreases with temperature. Molecular shape also affects viscosity.
Example: Water flows more easily than ethanol due to stronger hydrogen bonding and lower molar mass.
Capillary Action
Capillary action is the rise of liquid in a thin tube against gravity, caused by adhesive and cohesive forces. Adhesion pulls liquid up the wall; cohesion pulls the rest along. The effect is stronger in narrower tubes.
Vaporization and Vapor Pressure
Vaporization and Dynamic Equilibrium
Vaporization is the process of changing from liquid to gas when thermal energy overcomes intermolecular forces. In a closed container, vaporization and condensation reach dynamic equilibrium. The pressure exerted by vapor molecules is called vapor pressure.
Boiling Point: The temperature at which vapor pressure equals external pressure.
Enthalpy of Vaporization: The heat required to vaporize one mole of liquid ().
Sublimation and Fusion
Sublimation and Deposition
Sublimation is the process where molecules of a solid escape directly as gas when they gain sufficient energy. The reverse process is deposition.
Example: Dry ice (solid CO2) sublimates at room temperature.
Fusion (Melting) and Freezing
Fusion (melting) occurs when a solid is heated and molecules gain enough energy to overcome intermolecular forces, changing into a liquid. Freezing is the reverse process.
Heating Curve of Water
Segments of the Heating Curve
The heating curve of water illustrates temperature changes and phase transitions as heat is added. Key segments include warming ice, melting, warming liquid water, evaporating, and warming steam.
Phase Diagram
Understanding Phase Diagrams
A phase diagram shows the physical state of a substance at various temperatures and pressures. It includes regions for solid, liquid, and gas, lines for state changes, and special points like the triple point and critical point.
Triple Point: All three phases coexist.
Critical Point: Liquid and gas become indistinguishable.
Supercritical Fluid: Above critical temperature and pressure, exhibits properties of both gas and liquid.
Water: An Extraordinary Substance
Unique Properties of Water
Water is a liquid at room temperature due to strong hydrogen bonding. It is a universal solvent, has high specific heat, and expands upon freezing, making ice less dense than liquid water.
Example: Ice floats on water due to lower density.
