 Back
BackLiquids, Solids, and Intermolecular Forces: Key Concepts and Properties
Study Guide - Smart Notes

Liquids and Their Properties
Surface Tension
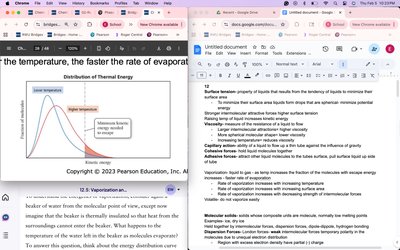
Surface tension is a property of liquids that results from the tendency of liquids to minimize their surface area. This phenomenon is due to intermolecular forces acting at the surface of the liquid.
Definition: Surface tension is the energy required to increase the surface area of a liquid by a unit amount.
Cause: Liquids form spherical drops to minimize potential energy and surface area.
Effect of Intermolecular Forces: Stronger intermolecular forces result in higher surface tension.
Temperature: Increasing the temperature raises the kinetic energy of molecules, reducing surface tension.
Viscosity
Viscosity is a measure of a liquid's resistance to flow, determined by the strength of intermolecular attractions and molecular shape.
Definition: Viscosity quantifies how easily a liquid flows.
Intermolecular Forces: Larger attractions lead to higher viscosity.
Molecular Shape: More spherical molecules have lower viscosity.
Temperature: Higher temperatures decrease viscosity.
Capillary Action
Capillary action is the ability of a liquid to flow up a thin tube against gravity, resulting from cohesive and adhesive forces.
Cohesive Forces: Hold liquid molecules together.
Adhesive Forces: Attract liquid molecules to the tube's surface, pulling the liquid upward.
Phase Changes and Energetics
Vaporization
Vaporization is the process by which a liquid becomes a gas. As temperature increases, more molecules have enough energy to escape the liquid phase.
Endothermic Process: Requires energy input.
Rate Factors: Increases with temperature, surface area, and decreases with stronger intermolecular forces.
Volatile Liquids: Vaporize easily due to weak intermolecular forces.
Heat of Vaporization
The heat of vaporization is the amount of energy required to vaporize one mole of a liquid at constant pressure.
Endothermic: Always positive.
Formula:
Condensation
Condensation is the reverse of vaporization, where a gas becomes a liquid. It is an exothermic process.
Relationship:
Vapor Pressure
Vapor pressure is the pressure exerted by a vapor in dynamic equilibrium with its liquid phase. It depends on the strength of intermolecular forces.
Weaker Forces: Higher vapor pressure.
Dynamic Equilibrium: Rate of vaporization equals rate of condensation.
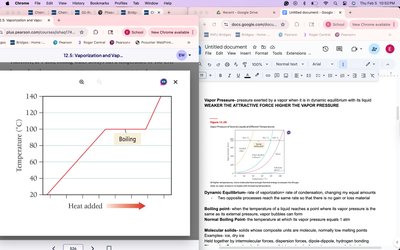
Boiling Point
The boiling point is the temperature at which a liquid's vapor pressure equals the external pressure, allowing vapor bubbles to form.
Normal Boiling Point: Temperature at which vapor pressure equals 1 atm.
At Boiling Point: Temperature remains constant as liquid converts to vapor.
Types of Solids and Intermolecular Forces
Molecular Solids
Molecular solids are composed of molecules held together by intermolecular forces, typically with low melting points.
Examples: Ice, dry ice.
Forces: Dispersion (London), dipole-dipole, hydrogen bonding.
Dispersion Forces (London Forces)
Dispersion forces are weak intermolecular forces arising from temporary polarity due to uneven electron distribution.
All molecules: Experience dispersion forces.
Strength: Increases with molar mass and electron cloud size.
Shape: More surface contact increases strength.
Dipole-Dipole Forces
Dipole-dipole forces exist between polar molecules with permanent dipoles.
Bond Polarity: Depends on electronegativity difference and molecular shape.
Miscibility: Polar substances mix well with other polar substances.
Hydrogen Bonding
Hydrogen bonding is the strongest type of intermolecular force, occurring when hydrogen is bonded to highly electronegative atoms (F, O, N).
Super Dipole-Dipole: Large partial charges.
Biological Importance: Found in DNA structure.
Ion-Dipole Forces
Ion-dipole forces occur when ionic compounds are mixed with polar compounds, important in solution chemistry.
Classification of Solids
Polymorphs
Polymorphs are different crystal structures that can exist for the same compound, resulting in varied properties.
Ionic Solids
Ionic solids are composed of ions held together by strong electrostatic (Coulombic) forces, typically with high melting points.
Example: Table salt (NaCl).
Coordination Number: Number of close cation-anion interactions; higher values indicate greater stability.
Rock Salt Structure: NaCl has a 1:1 ratio, with Cl ions in a face-centered cubic arrangement.
Atomic Solids
Atomic solids are composed of atoms and include nonbonding, metallic, and network covalent types.
Nonbonding Atomic Solids: Held by weak dispersion forces, low melting points.
Metallic Atomic Solids: Held by metallic bonds, variable melting points.
Network Covalent Solids: Held by covalent bonds, high melting points (e.g., diamond, graphite).
Crystalline vs. Amorphous Solids
Crystalline Solids: Highly ordered arrangement (ionic, molecular, covalent network, metals).
Amorphous Solids: Randomly arranged particles, no specific pattern.
Carbon Allotropes
Carbon exists in several crystalline forms, each with unique properties.
Graphite: Flat sheets of carbon atoms in hexagonal rings, good electrical conductor, slippery, sp2 hybridization.
Diamond: Tetrahedral geometry, sp3 hybridization, very hard, electrical insulator, thermal conductor.
Fullerenes (C60): Spherical clusters, held by dispersion forces.
Nanotubes: Cylindrical structures, strong, single or multi-walled.
Silicates
Silicates are network covalent atomic solids containing silicon, oxygen, and various metals.
Band Theory and Electrical Properties
Band Theory
Band theory explains bonding in atomic solids using molecular orbital theory, describing energy bands and gaps.
Valence Band: Formed from bonding molecular orbitals.
Conduction Band: Formed from antibonding molecular orbitals.
Band Gap: Energy difference between valence and conduction bands.
Electrical Conductivity
Insulators: Large band gap, no conductivity.
Semiconductors: Small band gap, conductivity increases with temperature.
Conductors: No band gap.
Doping: Adding impurities to increase conductivity (n-type and p-type semiconductors).
Polymers
Structure and Types
Polymers are long chain-like molecules composed of repeating units called monomers.
Natural Polymers: Found in living organisms (starch, proteins, DNA).
Synthetic Polymers: Made in laboratories (polyethylene, plastics, nylon).
Other Phase Changes
Sublimation and Deposition
Sublimation: Solid to gas.
Deposition: Gas to solid.
Fusion (Melting) and Freezing
Fusion (melting) occurs when a solid absorbs enough energy to overcome intermolecular forces and becomes a liquid. Freezing is the reverse process.
Melting Point: Temperature at which a solid becomes a liquid.
Heat of Fusion: Energy required to melt one mole of solid; always positive and less than heat of vaporization.
Crystallization:
