 Back
BackLiquids, Solids, and Intermolecular Forces: Mini-Textbook Study Guide
Study Guide - Smart Notes

Liquids, Solids, and Intermolecular Forces
Common States of Matter
The three most common states of matter are solid, liquid, and gas. Each state is characterized by the arrangement and movement of its constituent particles, and the boundaries between phases are called interfaces. A phase is a homogeneous part of a system separated by a well-defined boundary.
Solid phase: Molecules are locked in place by strong intermolecular forces, exhibiting long-range order.
Liquid phase: Molecules are mobile but maintain short-range order, spending time near their neighbors.
Gas phase: Molecules have complete freedom of movement and are highly disordered.
Plasma phase: A high-energy state where electrons are stripped from atoms (not common in general chemistry, but relevant in astrophysics).




Properties of States of Matter
The physical properties of solids, liquids, and gases differ due to the nature of their intermolecular forces and molecular motion.
Solid: High density, well-defined volume and shape, molecules vibrate and rotate around fixed positions.
Liquid: High density, well-defined volume but easily changes shape, molecules vibrate and rotate around positions that change over time.
Gas: Low density, fills the entire volume of any shape, molecules move randomly throughout the available space.
Phase Transitions
Changes between states of matter are called phase transitions. These include melting, freezing, vaporization, condensation, sublimation, and deposition. Phase transitions are driven by changes in temperature and pressure.
Melting (fusion): Solid to liquid
Freezing: Liquid to solid
Vaporization: Liquid to gas
Condensation: Gas to liquid
Sublimation: Solid to gas
Deposition: Gas to solid



Intermolecular Forces
Intermolecular forces are forces between molecules, while intramolecular forces hold atoms together within a molecule. Intermolecular forces are generally weaker than intramolecular forces but are crucial for holding condensed states (liquids and solids) together.
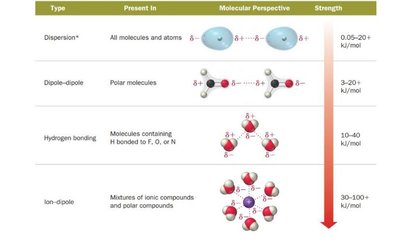
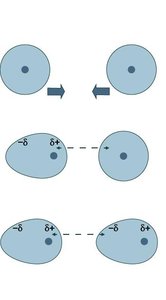
Dispersion (London) forces: Present in all molecules and atoms; arise from temporary dipoles due to electron motion.
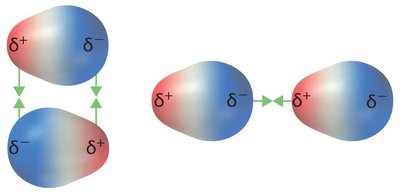
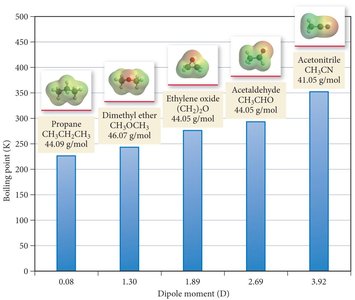
Dipole–dipole forces: Occur in polar molecules; molecules orient to maximize attractive interactions.
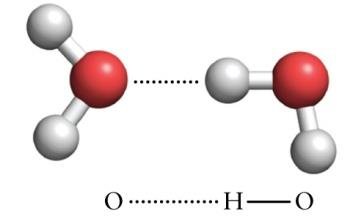
Hydrogen bonding: A strong dipole–dipole interaction involving H bonded to N, O, or F.
Ion–dipole forces: Attractive forces between ions and polar molecules.

Evidence of Intermolecular Forces
Strong intermolecular forces are evidenced by high boiling and melting points, large viscosity, and surface tension. For example, liquid nitrogen has weak intermolecular forces and low boiling/melting points, while liquid mercury has strong forces and high boiling/melting points.


Dispersion Forces and Molecular Properties
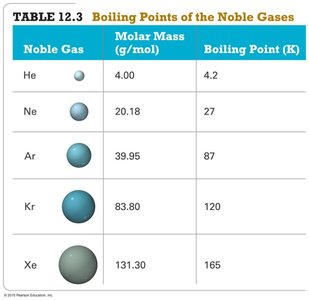
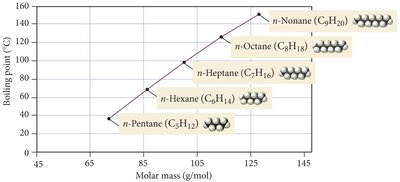
Dispersion forces increase with molecular size and shape. Larger electron clouds are more polarizable, leading to stronger dispersion forces and higher boiling points. Rod-like molecules have more contact points than spherical ones, increasing dispersion forces.

Hydrogen Bonding
Hydrogen bonds are unusually strong dipole–dipole interactions. They occur when hydrogen is bonded to highly electronegative atoms (N, O, F) and interacts with another electronegative atom. Hydrogen bonding leads to anomalously high boiling points and is crucial in biological molecules like DNA.
Donor: The molecule providing the hydrogen atom.
Acceptor: The molecule accepting the hydrogen bond.
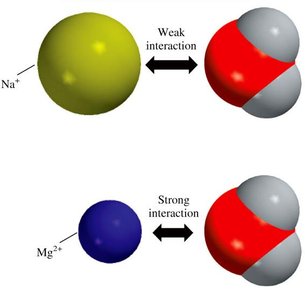
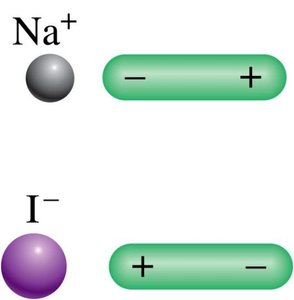
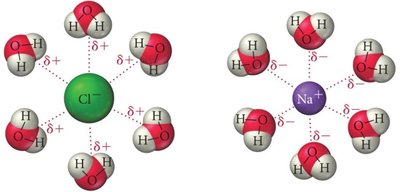
Ion–Dipole Forces and Hydration
Ion–dipole forces are attractive interactions between ions and polar molecules. These are important in the hydration of salts, where water molecules surround ions. Smaller cations have stronger ion–dipole interactions and hydrate more easily.
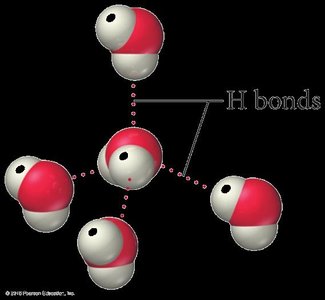
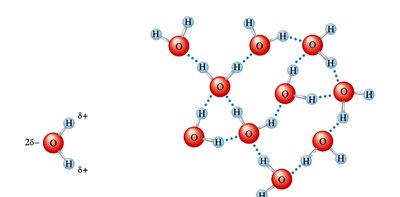
Hydrogen Bonds in Water and Ice
Hydrogen bonds are especially important in water and ice. Each water molecule can accept and donate hydrogen bonds, leading to unique properties such as lower density of ice compared to liquid water.
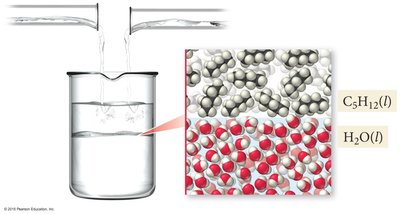
Immiscible Liquids
Nonpolar and polar liquids, such as pentane and water, are immiscible due to differences in intermolecular forces. Water molecules attract each other more strongly than they attract pentane molecules.
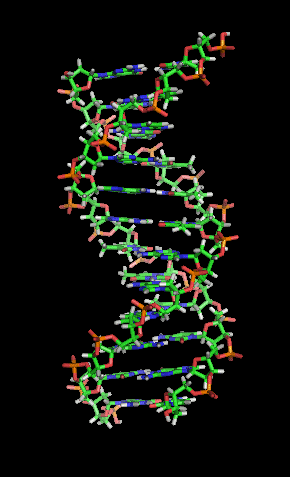
Hydrogen Bonding in DNA
Hydrogen bonds are responsible for pairing DNA strands. Guanine pairs with cytosine via three hydrogen bonds, and adenine pairs with thymine via two hydrogen bonds.
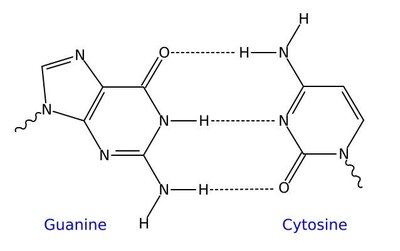
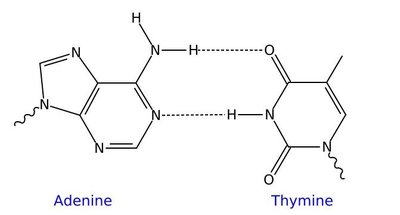
Surface Tension in Liquids
Surface tension is the energy required to increase the surface area of a liquid. Molecules at the surface experience a net inward force, causing droplets to adopt spherical shapes. Surface tension is responsible for phenomena such as water droplets on leaves and insects walking on water.



Phase Diagrams
A phase diagram graphically represents the regions and boundaries between conditions where a substance is stable as a solid, liquid, or gas. The triple point is where all three phases coexist. The critical point is the temperature and pressure above which a gas cannot be liquefied.
Fusion curve: Boundary between solid and liquid.
Evaporation curve: Boundary between liquid and gas.
Triple point: Unique set of conditions where all three phases coexist.
Critical point: Above this, the substance is a supercritical fluid.
Heats of Phase Transitions
The enthalpy changes associated with phase transitions are:
Heat of fusion (ΔHfus): Melting 1 mole of a solid at its melting point.
Heat of vaporization (ΔHvap): Vaporizing 1 mole of a liquid at its boiling point.
Heat of sublimation (ΔHsub): Subliming 1 mole of a solid.
From Hess's Law:
Heating Curve of Water
The heating curve of water shows the energy required to change water from ice to liquid to steam. The specific heat capacities differ for each phase:
Cice = 2.09 J g-1 °C-1
Cliquid = 4.18 J g-1 °C-1
Csteam = 2.01 J g-1 °C-1
Unique Properties of Water
Water has unique properties due to hydrogen bonding. Ice is less dense than liquid water, causing it to float. The structure of ice is a tetrahedral arrangement stabilized by hydrogen bonds.
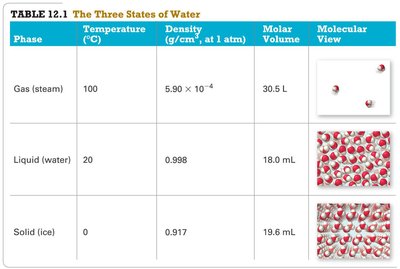
Summary Table: The Three States of Water
Phase | Temperature (°C) | Density (g/cm³, at 1 atm) | Molar Volume | Molecular View |
|---|---|---|---|---|
Gas (steam) | 100 | 5.90 × 10-4 | 30.5 L | Widely spaced molecules |
Liquid (water) | 20 | 0.998 | 18.0 mL | Close-packed, disordered molecules |
Solid (ice) | 0 | 0.917 | 19.6 mL | Ordered, close-packed molecules |
Summary Table: Types of Intermolecular Forces
Type | Present In | Molecular Perspective | Strength (kJ/mol) |
|---|---|---|---|
Dispersion | All molecules and atoms | Temporary dipoles | 0.05–20+ |
Dipole–dipole | Polar molecules | Permanent dipoles | 3–20 |
Hydrogen bonding | Molecules with H bonded to F, O, or N | Strong dipole–dipole | 10–40 |
Ion–dipole | Mixtures of ionic compounds and polar compounds | Ion and dipole interaction | 30–100+ |
Summary Table: Boiling Points of the Noble Gases
Noble Gas | Molar Mass (g/mol) | Boiling Point (K) |
|---|---|---|
He | 4.00 | 4.2 |
Ne | 20.18 | 27 |
Ar | 39.95 | 87 |
Kr | 83.80 | 120 |
Xe | 131.30 | 165 |
Additional info: This study guide expands on the original notes by providing definitions, examples, and context for each topic, ensuring completeness and academic quality for exam preparation.
