 Back
BackLiquids, Solids, and Intermolecular Forces: Structure and Properties
Study Guide - Smart Notes

Liquids, Solids, and Intermolecular Forces
Structure Determines Properties
The chemical composition and molecular structure of substances determine the type and strength of their intermolecular forces. These forces are responsible for holding liquids and solids together, and for the existence of condensed phases of matter. The phase (solid, liquid, or gas) depends on the magnitude of intermolecular forces relative to thermal energy.
Intermolecular forces: Attractive forces between molecules and atoms.
High thermal energy favors the gaseous state; low thermal energy favors liquid or solid states.
Properties of the Three Phases of Matter
Solids, liquids, and gases differ in shape, volume, and compressibility. Definite shape means matter keeps its shape in a container; indefinite shape means it takes the container's shape.
Solids, Liquids, and Gases: A Molecular Comparison
The density and molar volume of ice and liquid water are much closer to each other than to steam. Unusually, ice is less dense than liquid water, unlike most solids.
Solid: Definite shape and volume, incompressible.
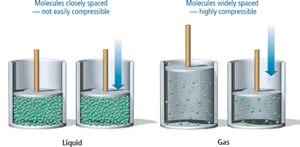
Liquid: Indefinite shape, definite volume, incompressible.
Gas: Indefinite shape and volume, compressible.
Liquids
Particles in a liquid are closely packed but can move around, making liquids incompressible and able to flow and take the shape of their container. However, they cannot expand to fill the container.

Explaining the Properties of Liquids
Higher density than gases due to close contact of particles.
Indefinite shape because particles can flow.
Definite volume due to limited freedom of movement.
Gases
Gas particles have complete freedom of motion, are widely spaced, and are highly compressible. Gases expand to fill their container, and their molar volume is much greater than that of solids or liquids.
Solids
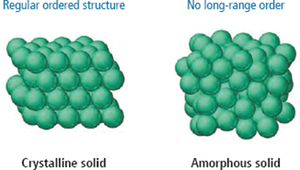
Solid particles are packed close together and fixed in position, making solids incompressible. They vibrate in place but do not flow. Solids can be crystalline (orderly geometric pattern) or amorphous (no long-range order).
Crystalline solids: Regular ordered structure (e.g., salt, diamond).
Amorphous solids: No long-range order (e.g., plastic, glass).
Changes Between States
Phase changes (solid, liquid, gas) can be induced by heating, cooling, or changing pressure.
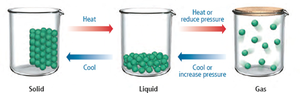
Intermolecular Forces
All attractive forces between particles are electrostatic in nature. The strength of these forces determines the state of matter and affects boiling and melting points. Stronger forces mean higher boiling and melting points.
Intermolecular forces are weaker than intramolecular (bonding) forces.
Types of intermolecular forces: dispersion, dipole–dipole, hydrogen bonding, ion–dipole.
Types of Intermolecular Forces
Dispersion forces: Temporary polarity due to unequal electron distribution; present in all molecules and atoms.
Dipole–dipole forces: Permanent polarity due to molecular structure; present in polar molecules.
Hydrogen bonding: Special dipole–dipole force when H is bonded to F, O, or N.
Ion–dipole forces: Attraction between ions and polar molecules; important in solutions.
Dispersion Forces
Dispersion forces (London forces) arise from temporary dipoles due to fluctuations in electron distribution. All molecules and atoms have dispersion forces, which increase with molar mass and surface contact.
Stronger dispersion forces lead to higher boiling points.
Molecular shape affects dispersion force strength.
Dipole–Dipole Attractions
Polar molecules have permanent dipoles, leading to dipole–dipole attractions. These forces raise boiling and melting points compared to nonpolar molecules of similar size.
Hydrogen Bonding
Hydrogen bonding occurs when H is bonded directly to a small, highly electronegative atom (F, O, or N). This results in strong intermolecular attraction, much stronger than regular dipole–dipole or dispersion forces, but weaker than covalent bonds.
Hydrogen bonds significantly increase boiling and melting points.
Ion–Dipole Attraction
Ion–dipole forces occur in mixtures of ionic compounds and polar molecules, determining solubility in water and other polar solvents.
Summary Table: Types of Intermolecular Forces
Type | Strength | Occurs In |
|---|---|---|
Dispersion | Weakest | All molecules/atoms |
Dipole–Dipole | Moderate | Polar molecules |
Hydrogen Bonding | Strong | H bonded to F, O, or N |
Ion–Dipole | Strongest | Ions in polar solvents |
Surface Tension
Surface tension is the tendency of liquids to minimize their surface area. It is higher for liquids with stronger intermolecular forces and decreases with increasing temperature.
Viscosity
Viscosity is the resistance of a liquid to flow. It increases with stronger intermolecular forces and decreases with higher temperature. Spherical molecules have lower viscosity.
Capillary Action and Meniscus
Capillary action is the ability of a liquid to flow up a thin tube against gravity, due to cohesive and adhesive forces. The meniscus is the curve at the liquid's surface, concave for water (adhesion > cohesion), convex for mercury (cohesion > adhesion).
Vaporization and Condensation
Vaporization is the process by which molecules escape from the liquid phase to the gas phase. It is endothermic, requiring energy input. Condensation is the reverse, exothermic process.
Rate of vaporization increases with temperature, surface area, and decreases with stronger intermolecular forces.
Volatile liquids evaporate easily; nonvolatile liquids do not.
Heat of Vaporization
The heat of vaporization () is the energy required to vaporize one mole of liquid. It is always endothermic and temperature-dependent.
Formula:
Dynamic Equilibrium and Vapor Pressure
In a closed container, vaporization and condensation reach dynamic equilibrium. The vapor pressure is the pressure exerted by the vapor in equilibrium with its liquid.
Higher vapor pressure means more volatile liquid.
Vapor pressure increases with temperature.
Boiling Point
The boiling point is the temperature at which the vapor pressure equals external pressure. Lower external pressure results in a lower boiling point.
Clausius–Clapeyron Equation
The Clausius–Clapeyron equation relates vapor pressure and temperature:
Two-point form:
Critical Point and Supercritical Fluids
At the critical temperature and pressure, the distinction between liquid and gas disappears, forming a supercritical fluid with properties of both.
Sublimation and Deposition
Sublimation is the transition from solid to gas without passing through the liquid phase. Deposition is the reverse process.
Melting (Fusion) and Freezing
Melting is the process of a solid becoming a liquid, requiring energy input (endothermic). Freezing is the reverse, exothermic process.
Heat of fusion (): Energy required to melt one mole of solid.
Heating Curve for Water
The heating curve shows temperature changes as water is heated, including plateaus at melting and boiling points where phase changes occur.
Phase Diagrams
Phase diagrams show the states of matter at various temperatures and pressures. Regions represent states; lines represent phase transitions. The triple point is where all three phases coexist. The critical point is the end of the vapor pressure curve.
Water: An Extraordinary Substance
Water is unique due to its hydrogen bonding, high boiling point, excellent solvent properties, high specific heat, and expansion upon freezing, making ice less dense than liquid water.
Boiling Points of Main-Group Hydrides
Hydrogen bonds in water result in a relatively high boiling point compared to other hydrides.
