 Back
BackLiquids, Solids, and Intermolecular Forces: Structured Study Notes
Study Guide - Smart Notes

Liquids, Solids, and Intermolecular Forces
Molecular Polarity
Molecular polarity is a fundamental concept in chemistry that describes the distribution of electrical charge over the atoms joined by a bond. The polarity of a molecule depends on both the polarity of its individual bonds and its overall shape.
Bond Polarity: Arises from unequal sharing of electrons between atoms due to differences in electronegativity.
Molecular Polarity: Refers to the polarity of the entire molecule, not just individual bonds.
Nonpolar Molecule: Any hydrocarbon or molecule with a symmetrical (perfect) shape. Examples include CCl4 and XeF4.
Perfect Shape I: Central atom has identical surrounding atoms and no lone pairs.
Perfect Shape II: Central atom has identical surrounding atoms and is linear or tetrahedral.
Polar Molecule: Any molecule whose Lewis Dot Structure does not have a perfect shape.
Example: BCl2F is polar due to its asymmetrical shape.
Intermolecular Forces
Intermolecular forces are the forces of attraction or repulsion between neighboring molecules. They are weaker than intramolecular forces (chemical bonds) but crucial for determining physical properties.
Intramolecular Forces: Exist within a molecule, bond atoms together, and influence chemical properties. Examples: ionic and covalent bonds.
Intermolecular Forces: Exist between molecules and influence physical properties such as boiling point, melting point, and solubility.
Types of Intermolecular Forces:
London Dispersion Forces (present in all molecules)
Dipole-Dipole Forces
Hydrogen Bonding
Ion-Dipole Forces
Ionic Forces
Example: The dominant force between MgSO4 and water is ion-dipole forces.
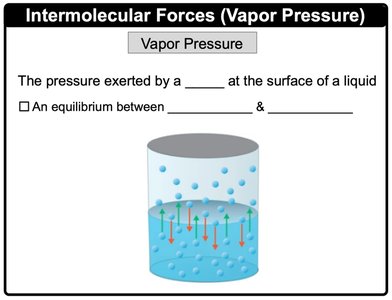
Vapor Pressure
Vapor pressure is the pressure exerted by a vapor at the surface of a liquid. It represents an equilibrium between evaporation and condensation.
Definition: The pressure exerted by a vapor at the surface of a liquid.
Equilibrium: Occurs between molecules leaving the liquid phase (evaporation) and returning to it (condensation).
Intermolecular Forces and Physical Properties
Intermolecular forces directly affect measurable physical properties such as boiling point, melting point, surface tension, and viscosity.
Boiling Point (BP): Temperature where vapor and liquid are in equilibrium.
Melting Point (MP): Temperature where solid and liquid are in equilibrium.
Surface Tension: Measure of cohesive forces on a liquid surface.
Viscosity: Resistance to flow; higher intermolecular forces lead to higher viscosity.
Direct Relationship: Stronger intermolecular forces result in higher boiling point, melting point, and surface tension.
Indirect Relationship: Stronger intermolecular forces result in lower vapor pressure.

Heating and Cooling Curves
Heating and cooling curves illustrate the amount of heat absorbed or released during phase changes. They are essential for understanding energy flow in substances.
Heating Curve: Endothermic process (heat absorbed).
Cooling Curve: Exothermic process (heat released).
Temperature Changes: Heat is converted to kinetic energy; temperature and average kinetic energy increase.
Phase Changes: Heat is converted to potential energy; temperature remains constant during phase change.
Formulas:
Specific Heat Capacity:
Enthalpy (Phase Change):
Total Energy:
Example: To convert 55.8 g of ice at -5 ºC to gas at 100 ºC, use the specific heat and enthalpy values for each phase.

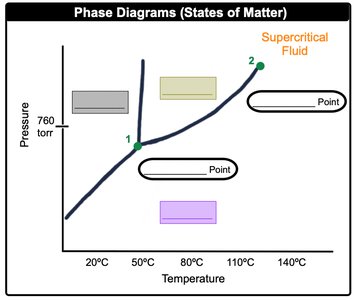
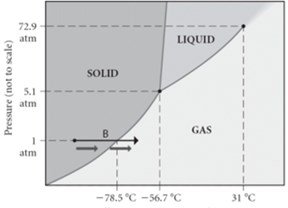
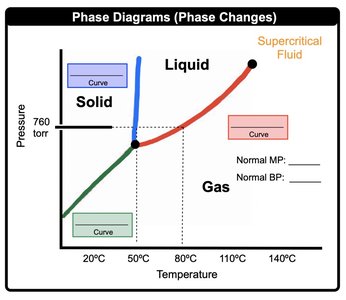
Phase Diagrams
Phase diagrams map the physical state of a pure substance as a function of pressure and temperature. They show regions of solid, liquid, gas, and supercritical fluid.
Triple Point: Unique set of conditions where all three states of matter are stable and in equilibrium.
Critical Point: Final set of pressure and temperature conditions where liquid and gas are indistinguishable.
Normal Pressure: Standard pressure of 1 atm or 760 mmHg.
Normal Melting Point: Solid to liquid transition at normal pressure.
Normal Boiling Point: Liquid to gas transition at normal pressure.
Clausius-Clapeyron Equation
The Clausius-Clapeyron equation relates the vapor pressure of liquids to temperature, allowing calculation of enthalpy of vaporization.
Linear Form: Used when a plot of ln P vs 1/T is given.
Where J/(mol·K), is vapor pressure, is enthalpy of vaporization, is temperature in K, and is a constant.
Two-Point Form: Used when two temperatures and/or two pressures are given.
Example: Calculate the vapor pressure of water at 60ºC using the two-point form.
Summary Table: Types of Intermolecular Forces
Type of Force | Present In | Relative Strength |
|---|---|---|
London Dispersion | All molecules | Weakest |
Dipole-Dipole | Polar molecules | Moderate |
Hydrogen Bonding | H bonded to N, O, F | Strong |
Ion-Dipole | Ions in polar solvents | Very strong |
Ionic | Ionic compounds | Strongest |
Additional info: Academic context and examples were added to clarify concepts and make the notes self-contained for exam preparation.
