 Back
BackLiquids, Solids, and Intermolecular Forces: Study Guide
Study Guide - Smart Notes

Liquids, Solids, and Intermolecular Forces
States of Matter
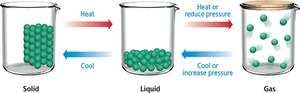
The three primary states of matter—solid, liquid, and gas—are distinguished by the arrangement and movement of their constituent particles. The physical properties of each state are determined by the balance between intermolecular forces and thermal energy.
Solid: Particles are closely packed and fixed in position, resulting in incompressibility and retention of shape.
Liquid: Particles are closely packed but can move past each other, making liquids incompressible but able to flow and take the shape of their container.
Gas: Particles are widely spaced and move freely, making gases highly compressible and able to expand to fill their container.
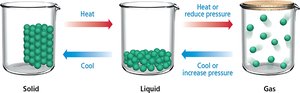
Phase Changes: Heating or cooling, as well as changes in pressure, can induce transitions between these states.
Example: Water can exist as ice (solid), liquid water, or water vapor (gas) depending on temperature and pressure.
Properties of Liquids
Liquids are characterized by closely packed particles with some freedom of movement. This allows liquids to flow and take the shape of their container, but not expand to fill it. Liquids are incompressible due to the close packing of their particles.
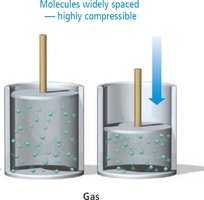
Properties of Gases
Gases have particles that are widely spaced and move freely, resulting in high compressibility and the ability to expand to fill their container. The molar volume of gases is much larger than that of solids or liquids.
Compressibility: Gases can be compressed by reducing the volume, bringing particles closer together.
Freedom of Motion: Gas particles move independently and rapidly.
Properties of Solids
Solids have particles packed closely together in fixed positions, making them incompressible and giving them a definite shape and volume. Solids do not flow.
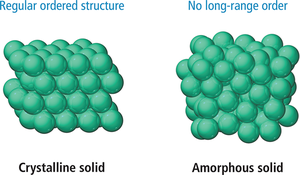
Crystalline Solids: Particles are arranged in an orderly geometric pattern (e.g., salt, diamond).
Amorphous Solids: Particles lack long-range order (e.g., plastic, glass).
Phase Changes
Phase changes occur when the kinetic energy of particles is altered or their freedom is limited. Heating causes solids to melt and liquids to boil, while cooling causes gases to condense and liquids to freeze. Pressure changes can also induce phase transitions.
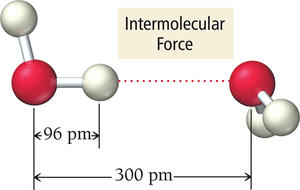
Intermolecular Forces
Intermolecular forces are attractive forces between molecules or atoms, responsible for the existence of condensed states (solids and liquids). These forces are much weaker than chemical bonds but play a crucial role in determining physical properties.
Types: Ion-ion, dipole-dipole, hydrogen bonding, dispersion forces.
Strength: Larger charges and shorter distances result in stronger attractions.
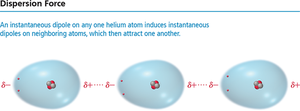
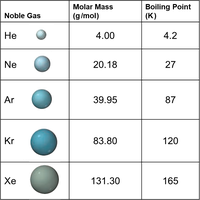
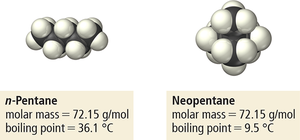
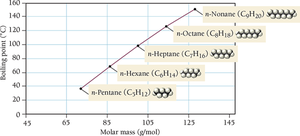
Dispersion Forces (London Forces)
Dispersion forces arise from temporary fluctuations in electron distribution, creating instantaneous dipoles that induce dipoles in neighboring molecules. These forces are present in all atoms and molecules and are the weakest type of intermolecular force.
Magnitude: Increases with molar mass and polarizability.
Shape: More surface-to-surface contact increases the strength of dispersion forces.
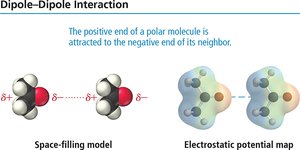
Dipole–Dipole Forces
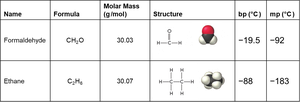
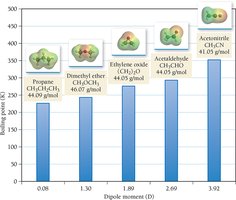
Dipole–dipole forces occur between polar molecules with permanent dipoles. These forces increase boiling and melting points compared to nonpolar molecules of similar size.
Dipole Moment: The measure of polarity in a molecule.
Example: CH2Cl2 exhibits dipole–dipole attractions due to its polar nature.

Solubility and Intermolecular Forces
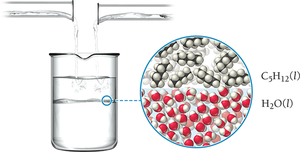
Solubility depends on the attractive forces between solute and solvent molecules. Polar substances dissolve in polar solvents, and nonpolar substances dissolve in nonpolar solvents. "Like dissolves like." Immiscible liquids do not mix due to differences in intermolecular forces.
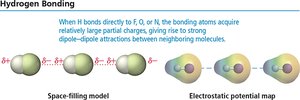
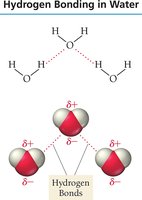
Hydrogen Bonding
Hydrogen bonding is a strong type of dipole–dipole interaction that occurs when hydrogen is bonded to highly electronegative atoms (O, N, F). The exposed proton attracts electron clouds from neighboring molecules, resulting in higher boiling and melting points.
Strength: Stronger than dipole–dipole or dispersion forces, but weaker than covalent bonds.
Example: Water and ethanol exhibit hydrogen bonding.
Ion–Dipole Forces
Ion–dipole forces occur in mixtures of ionic compounds and polar molecules. These forces are important for the solubility of ionic compounds in water and are the strongest intermolecular attractions in mixtures.
Summary Table: Types and Strengths of Intermolecular Forces
Type of Force | Relative Strength | Occurs Between |
|---|---|---|
Dispersion (London) | Weakest | All molecules/atoms |
Dipole–Dipole | Intermediate | Polar molecules |
Hydrogen Bonding | Strong (pure substances) | H bonded to O, N, or F |
Ion–Dipole | Strongest (mixtures) | Ions and polar molecules |
Surface Tension
Surface tension is the tendency of liquids to minimize their surface area due to cohesive forces. Liquids form spherical drops in the absence of gravity. The stronger the intermolecular forces, the higher the surface tension.
Viscosity
Viscosity is the resistance of a liquid to flow. Larger intermolecular attractions and less spherical molecular shapes increase viscosity.
Capillary Action and Meniscus
Capillary action is the ability of a liquid to flow up a thin tube against gravity, resulting from cohesive and adhesive forces. The meniscus is the curve at the liquid's surface in a tube, determined by the balance of these forces.
Vaporization and Condensation
Vaporization occurs when high-energy molecules escape the liquid phase. Condensation is the reverse process, where vapor molecules return to the liquid. The rate of evaporation increases with temperature and surface area.
Vapor Pressure
Vapor pressure is the pressure exerted by a vapor in equilibrium with its liquid. Higher vapor pressure indicates greater volatility. Vapor pressure increases with temperature.
Boiling Point
The boiling point is the temperature at which a liquid's vapor pressure equals external pressure. Lower external pressure results in a lower boiling point.
Sublimation and Deposition
Sublimation is the transition from solid to gas without passing through the liquid phase. Deposition is the reverse process. Both occur at temperatures below the melting point in closed containers.
Phase Diagrams
Phase diagrams show the states of matter and phase transitions at various temperatures and pressures. The critical point marks the formation of a supercritical fluid, and the triple point is where all three states coexist.
Water: An Extraordinary Substance
Water is unique due to its hydrogen bonding, high specific heat, and ability to expand upon freezing. It is an excellent solvent for ionic and polar substances and has a large dipole moment.
