 Back
BackLiquids, Solids, Intermolecular Forces, Solutions, and Chemical Kinetics: Mini-Study Guide
Study Guide - Smart Notes

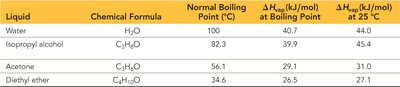
Liquids, Solids, and Intermolecular Forces
States of Matter and Their Properties
The physical state of a substance—gas, liquid, or solid—is determined by the strength of intermolecular forces relative to thermal energy. These forces influence density, shape, and compressibility.
Gas: Low density, indefinite shape and volume, weak intermolecular forces.
Liquid: High density, indefinite shape, definite volume, moderate intermolecular forces.
Solid: High density, definite shape and volume, strong intermolecular forces.
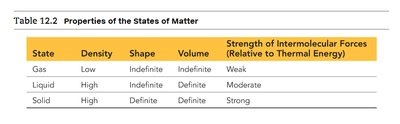
State | Density | Shape | Volume | Strength of Intermolecular Forces |
|---|---|---|---|---|
Gas | Low | Indefinite | Indefinite | Weak |
Liquid | High | Indefinite | Definite | Moderate |
Solid | High | Definite | Definite | Strong |
Types of Intermolecular Forces
Intermolecular forces are attractions between molecules and are weaker than intramolecular (covalent) bonds. The main types are:
Dispersion Forces: Present in all molecules, strongest in large, linear, non-polar molecules.
Dipole-Dipole Forces: Occur in polar molecules.
Hydrogen Bonds: Strongest, occur when H is bonded to F, O, or N.
Higher boiling points indicate stronger intermolecular forces.
Solutions and Solution Formation
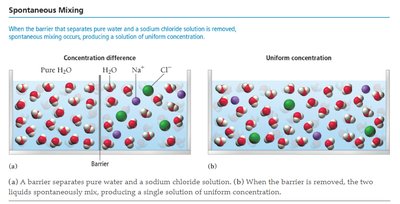
Spontaneous Mixing and Solution Formation
When two substances are mixed, spontaneous mixing occurs due to entropy, resulting in a homogeneous solution. The process depends on the relative strengths of solute-solvent, solute-solute, and solvent-solvent interactions.
Solvent-solute interactions > solute-solute and solvent-solvent: Solution forms.
Solvent-solute interactions = solute-solute and solvent-solvent: Solution forms.
Solvent-solute interactions < solute-solute and solvent-solvent: Solution may or may not form.
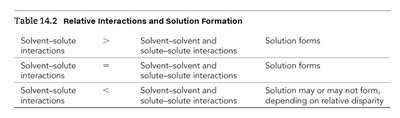
Solvent-solute interactions | Relative to Solvent-solvent and Solute-solute | Solution Formation |
|---|---|---|
> | Solvent-solvent and solute-solute | Solution forms |
= | Solvent-solvent and solute-solute | Solution forms |
< | Solvent-solvent and solute-solute | Solution may or may not form |
"Like dissolves like": Polar solvents dissolve polar solutes; non-polar solvents dissolve non-polar solutes.
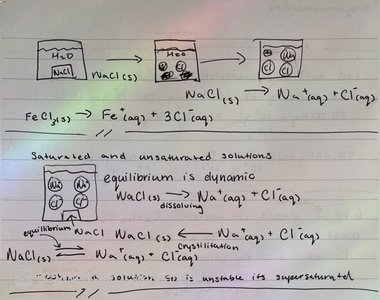
Saturated and Unsaturated Solutions
A saturated solution contains the maximum amount of solute that can dissolve at a given temperature. Unsaturated solutions can dissolve more solute. Equilibrium is dynamic, with dissolving and crystallization occurring simultaneously.
Saturated: Maximum solute dissolved; equilibrium between dissolving and crystallization.
Unsaturated: Less than maximum solute dissolved.
Supersaturated: More solute than equilibrium allows; unstable.
Concentration Units and Calculations
Common Units of Concentration
Concentration expresses the amount of solute in a given amount of solvent or solution. Common units include:
Molarity (M):
Molality (m):
Mole Fraction (X):
Percent by Mass (% m/m):
Parts per million (ppm):
Parts per billion (ppb):
Factors Affecting Solubility
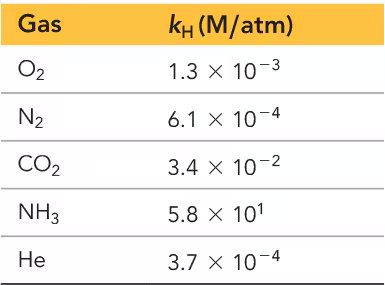
Henry's Law for Gas Solubility
The solubility of a gas in a liquid is proportional to the partial pressure of the gas above the solution, described by Henry's Law:
: Henry's law constant (units: M/atm)
: Partial pressure of the gas
Solubility increases with pressure for gases, but decreases with temperature.
Colligative Properties
Vapor Pressure Lowering, Freezing Point Depression, Boiling Point Elevation, Osmotic Pressure
Colligative properties depend only on the number of solute particles in solution, not their identity. Key properties include:
Vapor Pressure Lowering:
Freezing Point Depression:
Boiling Point Elevation:
Osmotic Pressure:
Where is the van't Hoff factor (number of particles), is molality, and are solvent-specific constants, is the gas constant, and is temperature in Kelvin.
Chemical Kinetics
Rate of Chemical Reaction
Chemical kinetics studies the speed of reactions and how they change with conditions. The rate is defined as the change in concentration of a reactant or product per unit time.
Rate: for reactants, for products
For a reaction :
Rate Laws and Reaction Order
The rate law expresses the relationship between reaction rate and concentrations of reactants:
General form:
is the rate constant; and are the orders with respect to A and B.
Orders are determined experimentally and do not necessarily match stoichiometric coefficients.
Zero, first, and second order reactions have distinct concentration-time profiles.

Example: Determining Reaction Order
By comparing initial rates and concentrations, the order of reaction can be deduced. For example, doubling the concentration of a reactant and observing the change in rate allows calculation of the order using:
Solve for to find the order.
Additional info: Rate laws are fundamental for predicting how changes in concentration affect reaction speed, and are essential for understanding mechanisms and designing chemical processes.
----------------------------------------
