 Back
BackMatter, Measurement, and Problem Solving: Foundations of General Chemistry
Study Guide - Smart Notes

Chapter 1: Matter, Measurement, and Problem Solving
Units of Measurement
Understanding and using the correct units is fundamental in chemistry. The International System of Units (SI) provides a standardized way to express measurements, ensuring consistency and clarity in scientific communication.
Standard Units: The SI system uses base units such as meter (m) for length, kilogram (kg) for mass, and second (s) for time.
Prefix Multipliers: Prefixes are used to represent multiples or fractions of base units, making it easier to express very large or very small quantities.
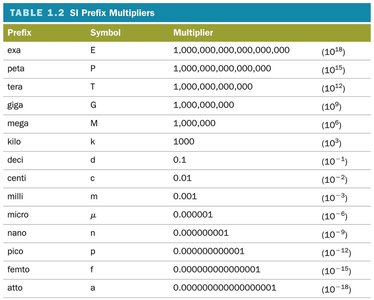
Example: 1 kilometer (km) = meters (m); 1 milligram (mg) = grams (g).
Derived Units
Derived units are combinations of base units that describe other physical quantities commonly encountered in chemistry.
Area: ; SI unit:
Volume: ; SI unit: , commonly or L in chemistry
Density: ; units: or
Speed: ; units:
Force: ; units: (newton, N)
Pressure: ; units: (pascal, Pa)
Energy: ; units: (joule, J)
Example: Calculating the area of a square with sides of 1 m:
Example: Calculating the volume of a cylinder:
Reliability of a Measurement
Measurements in chemistry must be both accurate and precise. The reliability of a measurement depends on the instrument used and the method of measurement.
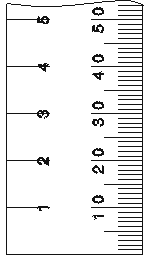
Making Measurements: Always record all certain digits and one estimated digit.
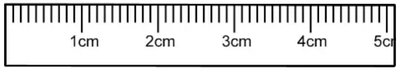
Example: Measuring with a ruler marked to the nearest 0.1 cm allows estimation to 0.01 cm.
Measuring Mass: Top-loading balances provide two decimal places (±0.01 g), while analytical balances provide four decimal places (±0.0001 g).


Measuring Volume: Read the volume at the bottom of the meniscus. Burets and graduated cylinders are common tools.

Significant Figures
Significant figures (sig figs) reflect the precision of a measured quantity. The rules for counting significant figures are essential for reporting and calculating with measured data.
Rule 1: All nonzero digits and zeros between them are significant. Leading zeros are not significant.
Rule 2: Trailing zeros after a decimal point are significant.
Rule 3: In scientific notation, only the digits in the mantissa are significant.
Rule 4: Trailing zeros in a number without a decimal point may or may not be significant; use scientific notation to clarify.
Exact Numbers: Numbers from counting, definitions, or constants have infinite significant figures and do not affect calculations.
Significant Figures in Calculations
When performing calculations, the number of significant figures in the result depends on the operation:
Addition/Subtraction: The result has the same number of decimal places as the measurement with the fewest decimal places.
Multiplication/Division: The result has the same number of significant figures as the measurement with the fewest significant figures.
Precision and Accuracy
Precision and accuracy are used to evaluate the quality of measurements:
Accuracy: How close a measurement is to the true value.
Precision: How close repeated measurements are to each other.
Random Error: Error with equal probability of being too high or too low; can be reduced by averaging.
Systematic Error: Error that consistently skews results in one direction; must be identified and corrected.
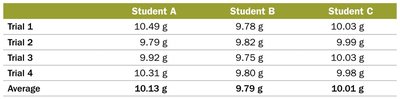
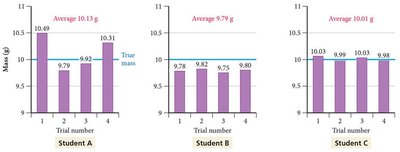
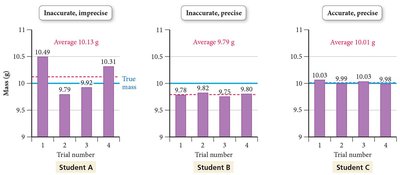
Central Tendency: Arithmetic mean (), median
Accuracy Evaluation: Absolute error (), relative error ()
Precision Estimate: Standard deviation (), relative standard deviation ()
Solving Chemical Problems
Problem-solving in chemistry often involves unit conversions and the use of equations. Dimensional analysis is a systematic approach to ensure correct units and results.
Unit Conversions: Use conversion factors to change from one unit to another. Always include units in calculations.
Dimensional Analysis: Multiply by conversion factors so that units cancel appropriately, leaving the desired unit.
Example: Converting 138 pm to cm:
Units Raised to a Power: When converting area or volume, conversion factors must be squared or cubed as appropriate.
Example:
Using Equations
Many chemical problems require solving equations for an unknown variable. Organize known and unknown quantities, select the appropriate equation, and solve algebraically.
Example: Finding the radius of a cylinder given volume and height:
Example: Calculating volume from mass and density:
Analyzing and Interpreting Data
Interpreting data, especially graphical data, is a key skill in chemistry. Always check the title, axes, and ranges before drawing conclusions from a graph.
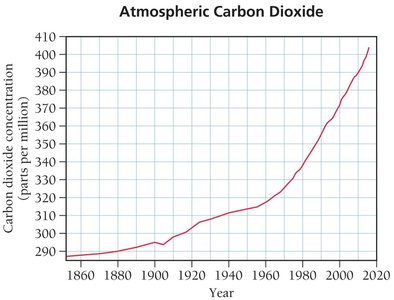
Example: Determining the concentration of CO2 in 1960 and 2010 from a graph.
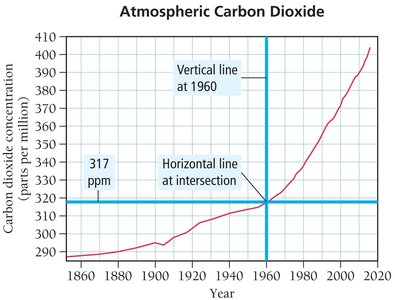
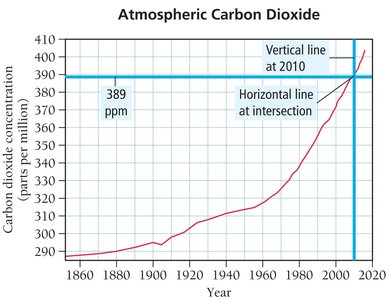
Calculating Change:
Average Rate of Increase:
Prediction: If the rate remains constant,
