 Back
BackMeasuring Enthalpy: Calorimetry and Heat Capacity
Study Guide - Smart Notes

Measuring Enthalpy and Calorimetry
Introduction to Enthalpy and Thermochemistry
Enthalpy (H) is a fundamental concept in thermochemistry, representing the total heat content of a system at constant pressure. The change in enthalpy (\( \Delta H \)) during a chemical reaction is crucial for understanding energy flow, predicting reaction spontaneity, and calculating energetic yields. Since absolute enthalpy values cannot be measured directly, chemists focus on determining enthalpy changes using indirect methods.
Enthalpy Change (\( \Delta H \)): Defined as \( \Delta H = H_{\text{final}} - H_{\text{initial}} \).
Importance: Helps determine if a reaction will occur spontaneously and how much energy is required or released.
Measurement Challenge: Instantaneous enthalpy values are not directly measurable; only changes can be determined.
Heat and Enthalpy at Constant Pressure
Under constant pressure, the heat transferred to or from a system (q) is equal to the enthalpy change (\( \Delta H \)). This relationship allows chemists to use heat measurements to estimate enthalpy changes in chemical processes.
Key Equation: At constant pressure, \( q = \Delta H \).
Variables to Measure: To determine heat transfer, one must measure the mass, temperature change, and heat capacity of the substances involved.
Heat Capacity and Specific Heat
Definitions and Concepts
Heat capacity and specific heat are essential properties that describe how substances respond to heat input. These properties are used to calculate the amount of energy required to change the temperature of a substance.
Heat Capacity (C): The amount of energy required to raise the temperature of a substance by 1 K (or 1 °C). Usually given for one mole of the substance (molar heat capacity).

Specific Heat Capacity (c or Cs): The amount of energy required to raise the temperature of 1 gram of a substance by 1 K (or 1 °C).
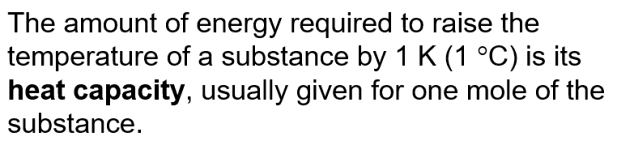
Typical Heat Capacities of Substances
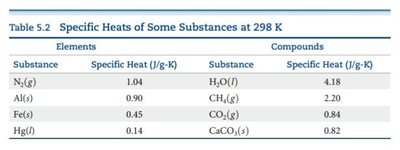
Different substances have different specific heat capacities, which influence how much their temperature changes when they absorb or release heat. Water, for example, has a high specific heat capacity, making it effective for thermal regulation.
Substance | Specific Heat (J/g·K) | Substance | Specific Heat (J/g·K) |
|---|---|---|---|
N2(g) | 1.04 | H2O(l) | 4.18 |
Al(s) | 0.90 | CH4(g) | 2.20 |
Fe(s) | 0.45 | CO2(g) | 0.84 |
Hg(l) | 0.14 | CaCO3(s) | 0.82 |

Calorimetry: Measuring Heat Changes
Constant Pressure Calorimetry (Coffee-Cup Calorimetry)
Constant pressure calorimetry is commonly used to measure the enthalpy change of reactions occurring in aqueous solution. The system is typically a simple calorimeter (such as nested Styrofoam cups), and the heat change is determined by measuring the temperature change of the water.
Specific Heat of Water: 4.184 J/g·K
Key Equation: where:
q = heat absorbed or released (J)
m = mass of solution (g)
Cs = specific heat capacity (J/g·K)
\( \Delta T \) = temperature change (K or °C)
Example Problem: Calculating Enthalpy Change
When 50.0 mL of 0.100 M AgNO3 and 50.0 mL of 0.100 M HCl are mixed in a constant-pressure calorimeter, the temperature increases from 22.30 °C to 23.11 °C. The reaction is:
AgNO3(aq) + HCl(aq) → AgCl(s) + HNO3(aq)
Calculate \( \Delta H \) for this reaction in kJ/mol AgNO3, assuming the combined solution has a mass of 100.0 g and a specific heat of 4.18 J/g·°C.

Bomb Calorimetry (Constant Volume Calorimetry)
Bomb calorimetry is used for reactions that are not suitable for coffee-cup calorimetry, such as combustion reactions. The reaction occurs in a sealed, rigid container (the bomb), and the heat absorbed or released by the water surrounding the bomb is measured. This provides a good approximation of the enthalpy change for the reaction.
Key Equation: where:
qrxn = heat of reaction (J)
Ccal = heat capacity of the calorimeter (J/K)
\( \Delta T \) = temperature change (K or °C)
Calibration: The calorimeter's heat capacity is determined in a preliminary run with a known energy output.

Summary Table: Comparison of Calorimetry Methods
Method | System | Measured Quantity | Equation | Typical Use |
|---|---|---|---|---|
Constant Pressure (Coffee-Cup) | Open to atmosphere | q (heat at constant P) | Reactions in solution | |
Bomb Calorimetry | Sealed, rigid container | q (heat at constant V) | Combustion reactions |
Key Takeaways
Enthalpy changes are central to understanding chemical reactions and energy flow.
Calorimetry allows for the indirect measurement of enthalpy changes by tracking heat transfer.
Heat capacity and specific heat are critical properties for calculating energy changes.
Different calorimetry methods are used depending on the reaction conditions and system constraints.
