 Back
BackModeling and Characterizing Chemical Reactions: Stoichiometry, Thermochemistry, Kinetics, and Equilibrium
Study Guide - Smart Notes

Unit Overview: Chemical Thinking and Chemical Change
This unit focuses on understanding how the structure and composition of substances determine their chemical reactivity and the outcomes of chemical processes. It introduces key concepts and methods for modeling chemical change, including stoichiometry, thermochemistry, kinetics, and equilibrium.
Essential Questions in Chemical Processes
Extent of Reaction: To what extent does a chemical process occur?
Reaction Rate: How fast does the process occur?
Quantities: How much substance is needed or produced?
Energy: How much energy is absorbed or released?
Major Modules in Characterizing Chemical Processes
Modeling Chemical Reactions: Identifying factors that affect chemical processes.
Understanding Proportions (Stoichiometry): Determining the amount of substance formed or consumed.
Tracking Energy (Thermochemistry): Predicting the amount of energy absorbed or released.
Modeling Chemical Reactions
Signature Features of Chemical Reactions
Chemical reactions involve reactants (initial substances) and products (new substances formed).
There is always a net transfer of energy between the system and its surroundings.
Reactions occur at varying speeds and extents; often, both reactants and products are present at equilibrium.
Atomic Rearrangement and Conservation Laws
During a chemical reaction, atoms are rearranged to form new particles with different compositions and structures.
The number of each type of atom and the total mass are conserved (Law of Conservation of Mass).
Example: The reaction of methane with oxygen:
Stoichiometric coefficients indicate the minimum proportions in which substances react, not the actual amounts present.
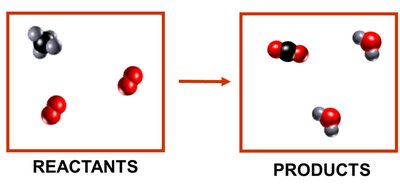
Limiting Reactant Concept
The limiting reactant is the substance that determines the maximum amount of product that can be formed.
Any other reactant present in excess will remain after the reaction is complete.
Example: In the combustion of methane, if oxygen is in excess, methane is the limiting reactant.
Stoichiometry: Quantitative Relationships in Chemical Reactions
Stoichiometry involves calculating the amounts of reactants and products in a chemical reaction using the balanced equation.
Relates the number of molecules, moles, or masses of substances involved.
Allows prediction of product yields and identification of limiting reactants.
Example: For the combustion of ethanol:
50 molecules of ethanol produce molecules of CO2.
Thermochemistry: Energy Changes in Chemical Reactions
Energy Transformations
Rearrangement of atoms during a reaction changes the potential energy of particles.
Energy may be released (exothermic) or absorbed (endothermic) by the system.
Example (Exothermic): Combustion of pentane:
Energy is released; products have lower potential energy than reactants.
Example (Endothermic): Photosynthesis:
Energy from sunlight is absorbed; products have higher potential energy.
Kinetics: Factors Affecting Reaction Rates
Collision Theory
For a reaction to occur, reactant particles must collide with sufficient energy and proper orientation.
The rate of reaction depends on:
Collision frequency: Increased by higher concentration, pressure, or temperature.
Configuration effectiveness: Proper orientation increases the likelihood of effective collisions.
Activation energy (Ea): The minimum energy required for a reaction to proceed.
Example: Increasing temperature increases both the speed and energy of particles, leading to more frequent and effective collisions.
Activation Energy and Catalysts
Lower activation energy means more particles can react, increasing the reaction rate.
Catalysts lower the activation energy without being consumed in the reaction.
Chemical Equilibrium: The Extent of Chemical Reactions
Dynamic Equilibrium
Many reactions are reversible; products can react to reform reactants.
At equilibrium, the rates of the forward and reverse reactions are equal, and concentrations of reactants and products remain constant.
Product-favored: More products than reactants at equilibrium. Reactant-favored: More reactants than products at equilibrium.
Factors Affecting Equilibrium
The extent of a reaction depends on the activation energy and configuration effectiveness of both forward and reverse reactions.
Temperature, pressure, and concentration changes can shift the position of equilibrium.
Summary Table: Factors Affecting Rate and Extent of Chemical Reactions
Factor | Effect on Rate | Effect on Extent (Equilibrium) |
|---|---|---|
Temperature | Increases collision frequency and energy, speeds up reaction | Can shift equilibrium position (direction depends on reaction type) |
Concentration/Pressure | Increases collision frequency, speeds up reaction | Shifts equilibrium toward side with fewer/more particles |
Activation Energy | Lower Ea increases rate | No direct effect on equilibrium position |
Configuration Effectiveness | Higher effectiveness increases rate | No direct effect on equilibrium position |
Catalyst | Lowers Ea, increases rate | No effect on equilibrium position |
Key Equations and Concepts
Balanced Chemical Equation:
Stoichiometric Calculations: Use mole ratios from the balanced equation to relate quantities of reactants and products.
Limiting Reactant: The reactant that is completely consumed first, limiting the amount of product formed.
Activation Energy (Ea): The minimum energy required for a reaction to proceed.
Chemical Equilibrium:
Visual Representations
Particulate-level diagrams are useful for visualizing the rearrangement of atoms during chemical reactions and for identifying reactants, products, and limiting reactants.

Energy diagrams illustrate the energy changes during reactions, showing whether a reaction is exothermic or endothermic and the activation energy barrier.
Conclusion
Understanding chemical change requires integrating concepts of stoichiometry, thermochemistry, kinetics, and equilibrium. By modeling chemical reactions, tracking proportions and energy, and analyzing the factors that affect rate and extent, chemists can predict and control chemical processes in both laboratory and real-world contexts.
