 Back
BackMolecules and Compounds: Polyatomic Ions and Compound Nomenclature
Study Guide - Smart Notes

Molecules and Compounds
Polyatomic Ions
Polyatomic ions are groups of covalently bonded atoms that carry an overall charge. These ions play a crucial role in the composition of many ionic compounds and are commonly encountered in general chemistry.
Definition: A polyatomic ion is a tightly bound group of two or more atoms that acts as a single charged entity.
Charge: The group as a whole has a net positive or negative charge.
Examples: Nitrate (NO3–), Sulfate (SO42–), Ammonium (NH4+).
Polyatomic Oxyanions
Oxyanions are negatively charged polyatomic ions containing oxygen. Their names and formulas follow specific patterns based on the number of oxygen atoms present.
Trioxides: Oxyanions with three oxygen atoms, typically ending in -ate (e.g., Nitrate, NO3–).
Tetraoxides: Oxyanions with four oxygen atoms, also often ending in -ate (e.g., Sulfate, SO42–).
Changing Oxygen Count: Decreasing the number of oxygens by one changes the ending from -ate to -ite (e.g., Nitrate NO3– to Nitrite NO2–).
Halogen Oxyanions
Halogen oxyanions are polyatomic ions containing a halogen and oxygen. Their names depend on the number of oxygen atoms attached to the halogen.
Base Name: Derived from the halogen (e.g., Chlor- for chlorine).
Prefixes and Suffixes: The number of oxygens determines the prefix/suffix:
# of Oxygens | Base Name |
|---|---|
4 | per____ate |
3 | ____ate |
2 | ____ite |
1 | hypo____ite |

Charge: All halogen oxyanions typically have a -1 charge.
Other Polyatomic Ions
Some polyatomic ions do not fit the standard naming patterns and must be memorized.
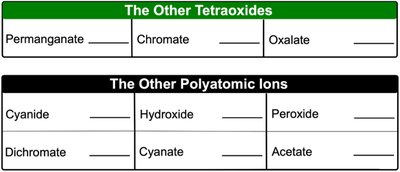
The Other Tetraoxides | |
|---|---|
Permanganate | MnO4– |
Chromate | CrO42– |
Oxalate | C2O42– |
The Other Polyatomic Ions | |
|---|---|
Cyanide | CN– |
Hydroxide | OH– |
Peroxide | O22– |
Dichromate | Cr2O72– |
Cyanate | OCN– |
Acetate | C2H3O2– or CH3COO– |
Polyatomic Cations
Ammonium (NH4+): The only common polyatomic ion with a +1 charge.
Mercury(I) (Hg22+): Composed of two mercury atoms bonded together.
Naming Ionic Compounds
Ionic compounds consist of a positive ion (cation) and a negative ion (anion). The rules for naming these compounds depend on the types of ions involved.
Cation: The metal or polyatomic cation is named first and keeps its elemental name.
Anion: The nonmetal or polyatomic anion is named second. If a nonmetal, its ending is changed to -ide.
Roman Numerals: Used for metals with more than one possible charge (e.g., Iron(III) chloride).
Polyatomic Ions: Always keep their standard names.
Writing Ionic Compound Formulas
To write the formula for an ionic compound, combine the cation and anion in ratios that balance the total positive and negative charges.
Step 1: Write the symbols and charges of the ions.
Step 2: Use subscripts to balance the charges so the total charge is zero.
Example: Magnesium sulfate:
Naming Ionic Hydrates
Ionic hydrates are ionic compounds that include water molecules in their structure. The number of water molecules is indicated by a prefix.
Hydrate: The water portion is named with a Greek prefix (mono-, di-, tri-, etc.) followed by "hydrate".
Example: is called copper(II) sulfate pentahydrate.
Naming Acids
Acids are compounds that typically begin with hydrogen. Their names depend on whether they are binary acids or oxyacids.
Binary Acids
Composition: Hydrogen and a nonmetal.
Naming: Use the prefix "hydro-", the base name of the nonmetal, and the suffix "-ic acid" (e.g., HCl is hydrochloric acid).
Oxyacids
Composition: Hydrogen, oxygen, and another element (usually a nonmetal).
Naming: If the polyatomic ion ends in "-ate", change to "-ic acid"; if it ends in "-ite", change to "-ous acid" (e.g., H2SO4 is sulfuric acid, H2SO3 is sulfurous acid).
Naming Molecular (Covalent) Compounds
Molecular compounds are formed between nonmetals. Numerical prefixes indicate the number of each atom present.
First Element: Named with its full name; prefix omitted if only one atom.
Second Element: Named with a prefix and the ending changed to "-ide".
Prefixes: mono-, di-, tri-, tetra-, penta-, hexa-, hepta-, octa-, nona-, deca-.
Example: CO2 is carbon dioxide; N2O4 is dinitrogen tetroxide.
Summary Table: Common Polyatomic Ions
Name | Formula | Charge |
|---|---|---|
Nitrate | NO3 | – |
Sulfate | SO4 | 2– |
Phosphate | PO4 | 3– |
Ammonium | NH4 | + |
Hydroxide | OH | – |
Carbonate | CO3 | 2– |
Acetate | C2H3O2 | – |
Additional info: The above notes are based on standard general chemistry curriculum and fill in logical gaps from the provided materials for completeness and clarity.
