 Back
BackMolecules and Compounds: Structure, Nomenclature, and Calculations
Study Guide - Smart Notes

Molecules and Compounds
Classification of Matter
Matter can be classified based on its composition and structure. Pure substances are divided into elements and compounds, which can be further categorized by their atomic or molecular nature.
Elements: Substances consisting of only one type of atom. They can exist as single atoms (atomic) or as molecules (molecular elements).
Compounds: Substances composed of two or more different elements chemically bonded together. Compounds can be molecular (formed by covalent bonds) or ionic (formed by ionic bonds).
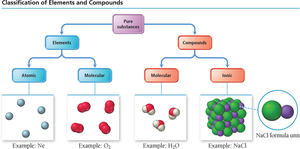
Elements: Atomic and Molecular
Elements can exist as individual atoms or as molecules made up of the same type of atom.
Atomic Elements: Exist as single atoms (e.g., Ne, Na, K).
Molecular Elements: Exist as molecules, often as diatomic (Br2, I2, N2, Cl2, H2, O2, F2) or polyatomic (P4, S8) species.
Chemical Bonds and Types of Compounds
Chemical Bonds
Atoms form compounds by bonding through the attraction between electrons and protons. The two main types of chemical bonds are:
Ionic Bonds: Formed by the transfer of electrons from metals to nonmetals, resulting in cations and anions held together by electrostatic forces.
Covalent Bonds: Formed by the sharing of electrons between two or more nonmetals, resulting in molecular compounds.
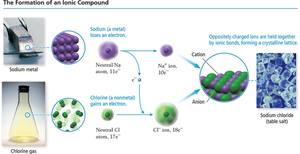
Ionic Bonds and Compounds
Ionic compounds are formed when electrons are transferred from a metal to a nonmetal. The resulting oppositely charged ions are attracted to each other, forming a crystalline lattice.
Process: Metal loses electrons to form a cation; nonmetal gains electrons to form an anion.
Example: Sodium (Na) reacts with chlorine (Cl) to form sodium chloride (NaCl).
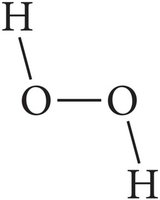
Covalent Bonds and Molecular Compounds
Covalent bonds involve the sharing of electrons between nonmetals, resulting in the formation of molecules. The term "molecule" specifically refers to compounds held together by covalent bonds.
Example: Water (H2O), carbon dioxide (CO2).
Representing Compounds: Formulas and Models
Chemical Formulas
Chemical formulas use element symbols and subscripts to indicate the types and numbers of atoms in a compound.
Empirical Formula: Shows the simplest whole-number ratio of atoms (e.g., HO for hydrogen peroxide).
Molecular Formula: Shows the actual number of atoms in a molecule (e.g., H2O2 for hydrogen peroxide).
Structural Formula: Shows how atoms are bonded in a molecule using lines for covalent bonds.
Molecular Models
Molecular models provide a three-dimensional representation of molecules, helping to visualize their shapes and the arrangement of atoms.
Ball-and-Stick Model: Atoms are represented as balls and bonds as sticks.
Space-Filling Model: Atoms fill the space between each other, more closely representing the actual appearance of a molecule.
Atoms are color-coded for clarity.

Nomenclature of Compounds
Naming Ionic Compounds
Ionic compounds are named based on the cation and anion present. The naming conventions depend on whether the ions are monoatomic or polyatomic, and whether the metal can form more than one type of ion.
Monoatomic Ions: Metal cations are named as the element followed by "ion" (e.g., Na+ is sodium ion).
Transition Metals: Use Roman numerals to indicate the charge (e.g., Fe2+ is iron(II) ion).
Nonmetal Anions: Named by adding "-ide" to the element stem (e.g., O2- is oxide ion).
Polyatomic Ions: Have specific names (e.g., NO3- is nitrate, SO42- is sulfate).
Writing Chemical Formulas for Ionic Compounds
Formulas are written so that the total positive and negative charges balance to zero. The crossover method is often used for ions with different charges.
Crossover Method: The charge of the cation becomes the subscript of the anion and vice versa, omitting the charge signs.
For polyatomic ions, use parentheses if more than one is needed.

Naming Molecular Compounds
Molecular (covalent) compounds are named using Greek prefixes to indicate the number of each type of atom present. The first element uses a prefix only if there is more than one atom; the second element always uses a prefix and ends with "-ide."
Examples: CO is carbon monoxide, PCl3 is phosphorus trichloride, Cl2O7 is dichlorine heptaoxide.
Atoms | Prefix | Atoms | Prefix |
|---|---|---|---|
1 | mono- | 6 | hexa- |
2 | di- | 7 | hepta- |
3 | tri- | 8 | octa- |
4 | tetra- | 9 | nona- |
5 | penta- | 10 | deca- |

Acids and Hydrates
Acids: Nomenclature and Formulas
Acids are compounds that release hydrogen ions (H+) in aqueous solution. Their names depend on the anion present:
Binary Acids: Use the prefix "hydro-" and the suffix "-ic acid" (e.g., HCl is hydrochloric acid).
Oxoacids: If the anion ends in "-ate," the acid ends in "-ic acid" (e.g., HNO3 is nitric acid); if the anion ends in "-ite," the acid ends in "-ous acid" (e.g., HNO2 is nitrous acid).
Hydrates
Hydrates are ionic compounds that contain a specific number of water molecules bound to them. The number of water molecules is indicated by Greek prefixes (e.g., FeSO4·H2O is iron(II) sulfate monohydrate).
Molar Mass, Mole Calculations, and Percent Composition
Molecular Weight, Formula Weight, and Molar Mass
The molecular weight is the sum of atomic weights of all atoms in a molecule (used for molecular compounds), while the formula weight is used for ionic compounds. The molar mass is the mass of one mole of a substance, numerically equal to the molecular or formula weight, with units of g/mol.
Example: The molar mass of NaCl is 58.44 g/mol.
Mole Calculations
Mole calculations use the molar mass as a conversion factor between grams and moles, and Avogadro's number () as a conversion factor between moles and number of particles.
Key Equations:
Mass Percent Composition
Mass percent composition expresses the percentage by mass of each element in a compound.
Equation:
Empirical and Molecular Formulas
Empirical and Molecular Formulas
The empirical formula gives the simplest whole-number ratio of atoms in a compound, while the molecular formula gives the actual number of atoms. The molecular formula is always a whole-number multiple of the empirical formula.
Equation:
where
Combustion Analysis
Combustion analysis is used to determine the empirical formula of compounds containing carbon, hydrogen, and oxygen by burning the sample and measuring the amounts of CO2 and H2O produced. Advanced instrumental methods such as ICP-MS (Inductively Coupled Plasma Mass Spectrometry), AAS (Atomic Absorption Spectroscopy), and AES (Atomic Emission Spectroscopy) are also used for elemental analysis.
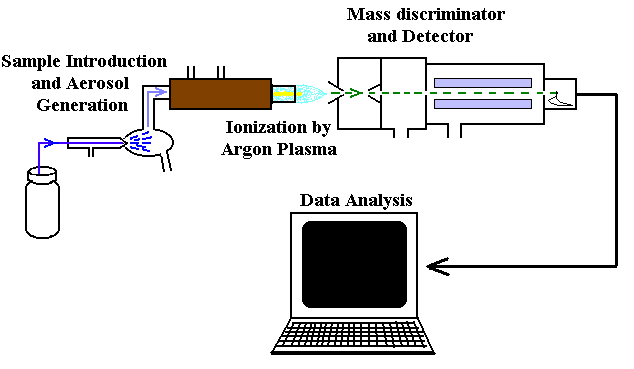
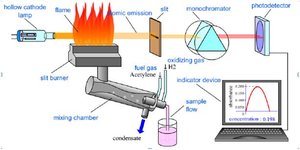
Additional info: The images for ICP-MS and AAS/AES are included as they directly relate to the instrumental methods mentioned for combustion analysis and elemental determination.
